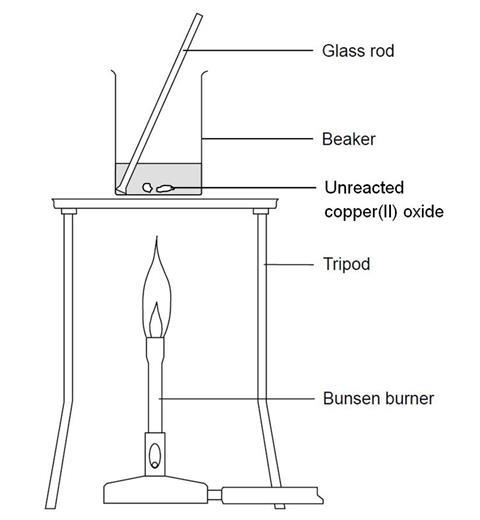
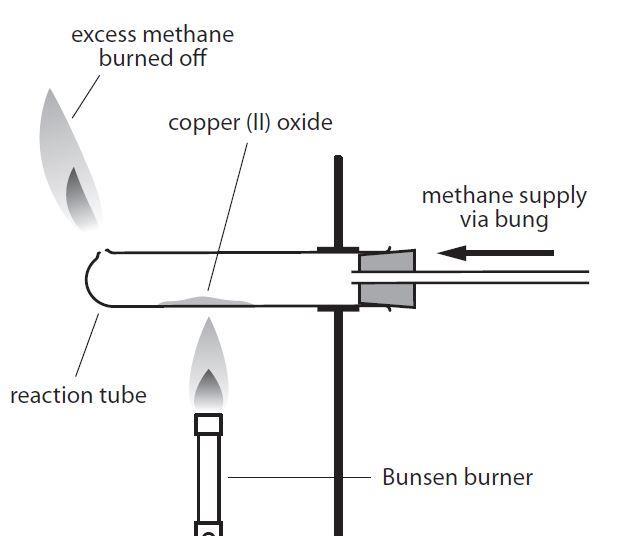
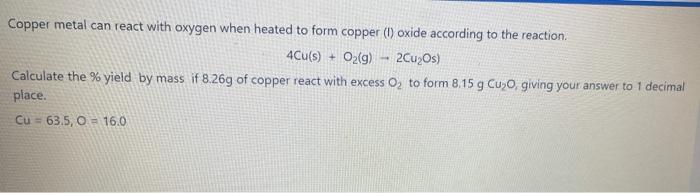When ammonia is passed over heated copper oxide, the metallic coper is obtained. The reaction shows - YouTube
What is the observation when aqueous hydrogen peroxide is shaken with copper(ll) oxide in a test tube? - Quora

Write balanced chemical equations for the following word equation:Copper + Oxygen → Copper (II) oxide

SOLVED: There are two steps in the extraction of copper metal from chalcocite, a copper ore. In the first step, copper(I) sulfide and oxygen react to form copper(I) oxide and sulfur dioxide:

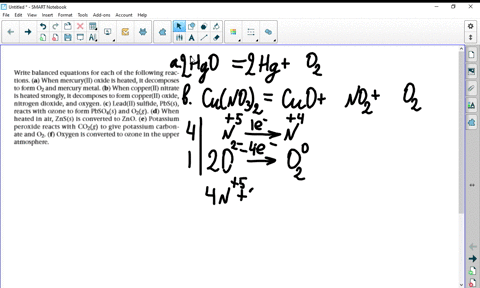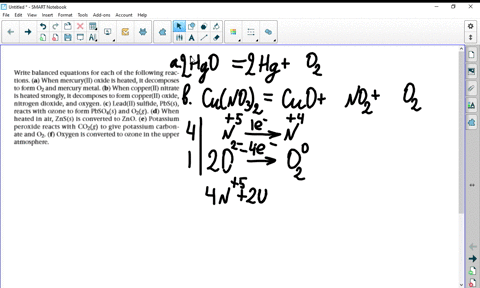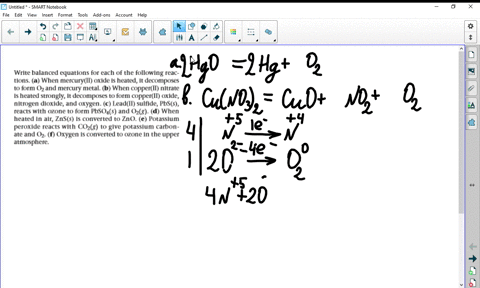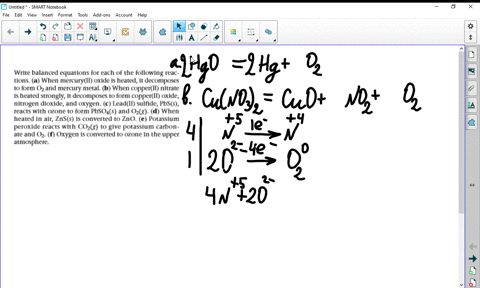
a) Example the folloeing in terms of gain or loss of oxygen with one example each : (b) When copper powder is heated strongly in air, it forms copper oxide. Write a

Write balanced chemical equations for the following word equation:Copper + Oxygen → Copper (II) oxide

SOLVED: 2. A sample of copper metal weighing 2.50 grams is heated in the presence of oxygen gas to form a copper oxide. After the reaction is complete, the final mass f

SOLVED: A student ix performing an experiment in the laboratory to determine the oxidation of copper (Cu): Cu reacts with oxygen in the air to form CuO in the following reaction: Cu (