![A Cycloisomerization/Friedel–Crafts Alkylation Strategy for the Synthesis of Pyrano[3,4-b]indoles | Organic Letters A Cycloisomerization/Friedel–Crafts Alkylation Strategy for the Synthesis of Pyrano[3,4-b]indoles | Organic Letters](https://pubs.acs.org/cms/10.1021/ol201532k/asset/images/ol201532k.social.jpeg_v03)
A Cycloisomerization/Friedel–Crafts Alkylation Strategy for the Synthesis of Pyrano[3,4-b]indoles | Organic Letters

Synthesis of Phenolic Coumestans via a Sequential Dehydrogenation/Oxa-Michael Addition Reaction of 2′,4′-Dihydroxyl-3-arylcoumarins | The Journal of Organic Chemistry
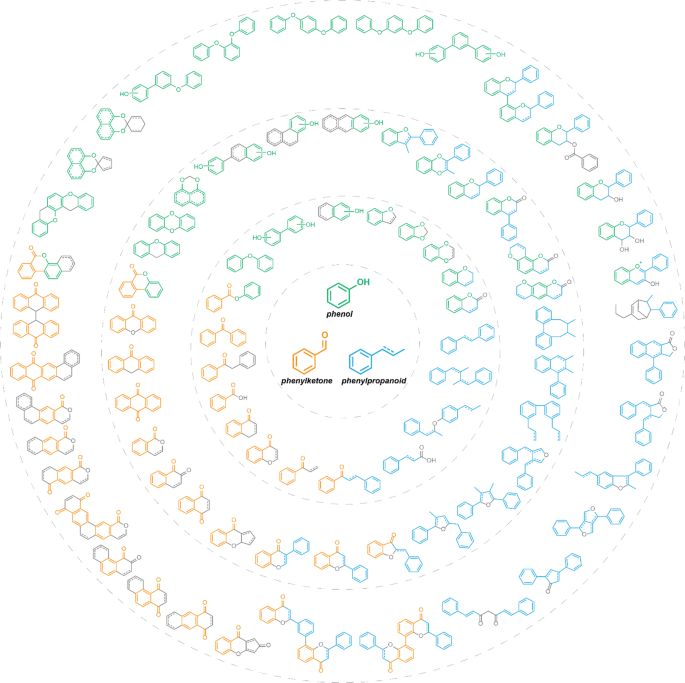
Structural derivatization strategies of natural phenols by semi-synthesis and total-synthesis | SpringerLink
![PDF] Synthesis and antimicrobial activity of 5-aminoquinoline and 3-amino phenol derivatives | Semantic Scholar PDF] Synthesis and antimicrobial activity of 5-aminoquinoline and 3-amino phenol derivatives | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/4fad0b44f869fcfe1ccdc093a2a1e670d3a4bdbb/2-Table1-1.png)
PDF] Synthesis and antimicrobial activity of 5-aminoquinoline and 3-amino phenol derivatives | Semantic Scholar

Calixarene‐Type Macrocycles by Oxidation of Phenols Related to Vitamin E - Rosenau - 2002 - Angewandte Chemie International Edition - Wiley Online Library
Structural derivatization strategies of natural phenols by semi-synthesis and total-synthesis | SpringerLink
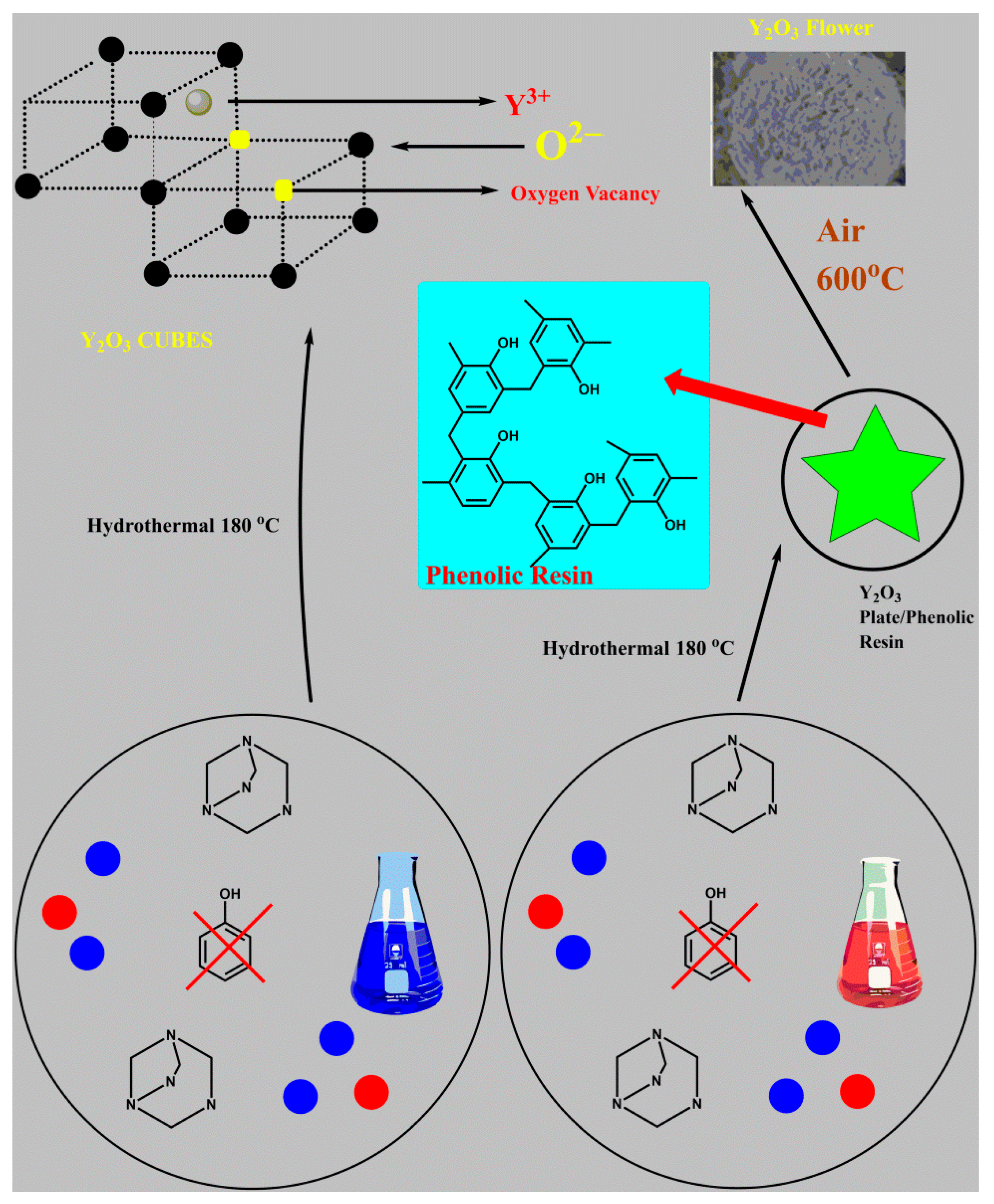
Applied Sciences | Free Full-Text | Yttrium Oxide Nanoparticle Synthesis: An Overview of Methods of Preparation and Biomedical Applications
Structural derivatization strategies of natural phenols by semi-synthesis and total-synthesis | SpringerLink
![Enantioselective Synthesis of Dihydrospiro[indoline-3,4′-pyrano[2,3-c]pyrazole] Derivatives via Michael/Hemiketalization Reaction | Organic Letters Enantioselective Synthesis of Dihydrospiro[indoline-3,4′-pyrano[2,3-c]pyrazole] Derivatives via Michael/Hemiketalization Reaction | Organic Letters](https://pubs.acs.org/cms/10.1021/acs.orglett.6b00287/asset/images/medium/ol-2016-00287p_0001.gif)
Enantioselective Synthesis of Dihydrospiro[indoline-3,4′-pyrano[2,3-c]pyrazole] Derivatives via Michael/Hemiketalization Reaction | Organic Letters
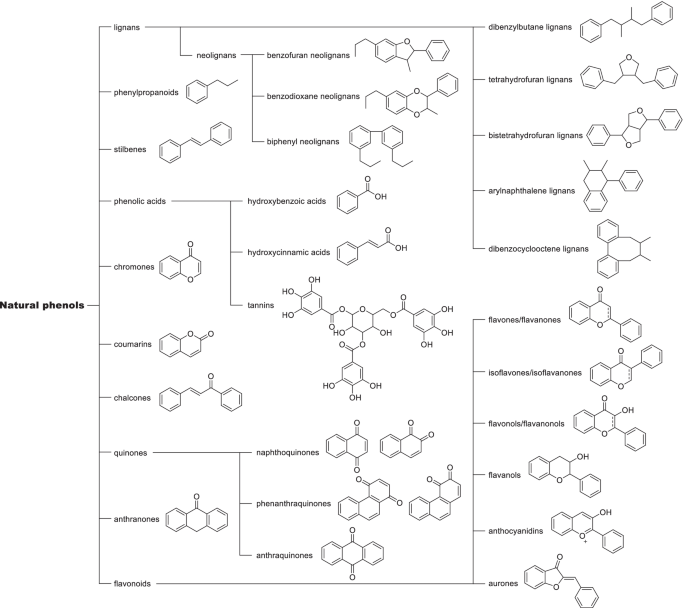
Structural derivatization strategies of natural phenols by semi-synthesis and total-synthesis | SpringerLink
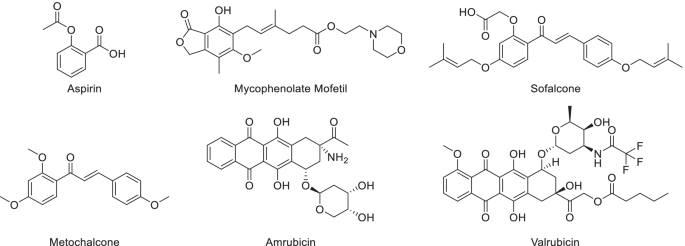
![Substituted 1,1'-biphenyl Compounds, Analogues Thereof, And Methods Using Same Bi; Yingzhi ; et al. [Arbutus Biopharma Corporation] Substituted 1,1'-biphenyl Compounds, Analogues Thereof, And Methods Using Same Bi; Yingzhi ; et al. [Arbutus Biopharma Corporation]](https://uspto.report/patent/app/20210052585/US20210052585A1-20210225-C00002.png)
Substituted 1,1'-biphenyl Compounds, Analogues Thereof, And Methods Using Same Bi; Yingzhi ; et al. [Arbutus Biopharma Corporation]

Biosynthesis pathway of phenolic acids and stilbene from phenylalanine. | Download Scientific Diagram

Bis(amino)cyclopropenium Ion as a Hydrogen-Bond Donor Catalyst for 1,6-Conjugate Addition Reactions | The Journal of Organic Chemistry
![PDF) Dibutylamine (DBA): A highly efficient catalyst for Synthesis of pyrano[2,3-d]pyrimidine derivatives in aqueous media PDF) Dibutylamine (DBA): A highly efficient catalyst for Synthesis of pyrano[2,3-d]pyrimidine derivatives in aqueous media](https://www.researchgate.net/profile/Ajmal-Bhat/publication/277726791/figure/tbl1/AS:669708953059335@1536682465183/Synthesis-of-pyrano2-3-dpyrimidine-derivatives-4a-j_Q320.jpg)
PDF) Dibutylamine (DBA): A highly efficient catalyst for Synthesis of pyrano[2,3-d]pyrimidine derivatives in aqueous media

Synthesis of Indolequinones from Bromoquinones and Enamines Mediated by Cu(OAc)2·H2O | The Journal of Organic Chemistry

Diketopyrrolopyrroles: Synthesis, Reactivity, and Optical Properties - Grzybowski - 2015 - Advanced Optical Materials - Wiley Online Library
![Intramolecular Diels–Alder Cycloaddition of Furan-Derived β-Enamino Diketones: An Entry to Diastereoselective Synthesis of Polycyclic Pyrano[3,2-c]quinolin-5-one Derivatives | The Journal of Organic Chemistry Intramolecular Diels–Alder Cycloaddition of Furan-Derived β-Enamino Diketones: An Entry to Diastereoselective Synthesis of Polycyclic Pyrano[3,2-c]quinolin-5-one Derivatives | The Journal of Organic Chemistry](https://pubs.acs.org/cms/10.1021/acs.joc.1c03163/asset/images/medium/jo1c03163_0010.gif)
Intramolecular Diels–Alder Cycloaddition of Furan-Derived β-Enamino Diketones: An Entry to Diastereoselective Synthesis of Polycyclic Pyrano[3,2-c]quinolin-5-one Derivatives | The Journal of Organic Chemistry
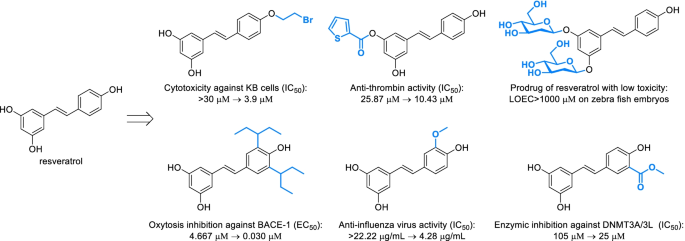
Structural derivatization strategies of natural phenols by semi-synthesis and total-synthesis | SpringerLink

Rhodium(I)-Catalyzed Cyclization Reaction of o-Alkynyl Phenols and Anilines. Domino Approach to 2,3-Disubstituted Benzofurans and Indoles | Organic Letters
Foods | Free Full-Text | A Comprehensive Survey of Phenolic Constituents Reported in Monofloral Honeys around the Globe

Trisubstituted Aziridine Ring-Opening by Phenol Derivatives: Stereo- and Regioselective Formation of Chiral Tertiary Alkyl-Aryl Ethers | Organic Letters

Synthesis of Phenolic Coumestans via a Sequential Dehydrogenation/Oxa-Michael Addition Reaction of 2′,4′-Dihydroxyl-3-arylcoumarins | The Journal of Organic Chemistry
![EtO)3Si(CH2)3NH3+][CH3COO−] as a novel basic ionic liquid catalyzed green synthesis of new 2-(phenylsulfonyl)-1H-benzo[a]pyrano[2,3-c]phenazin-3-amine derivatives - ScienceDirect EtO)3Si(CH2)3NH3+][CH3COO−] as a novel basic ionic liquid catalyzed green synthesis of new 2-(phenylsulfonyl)-1H-benzo[a]pyrano[2,3-c]phenazin-3-amine derivatives - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0022286022002319-ga1.jpg)


