ADDING MULTIMEDIA Teva Announces FDA Approval of First and Only Digital Inhaler with Built-In Sensors – ProAir® Digihaler™ (albuterol sulfate 117 mcg) Inhalation Powder | Business Wire
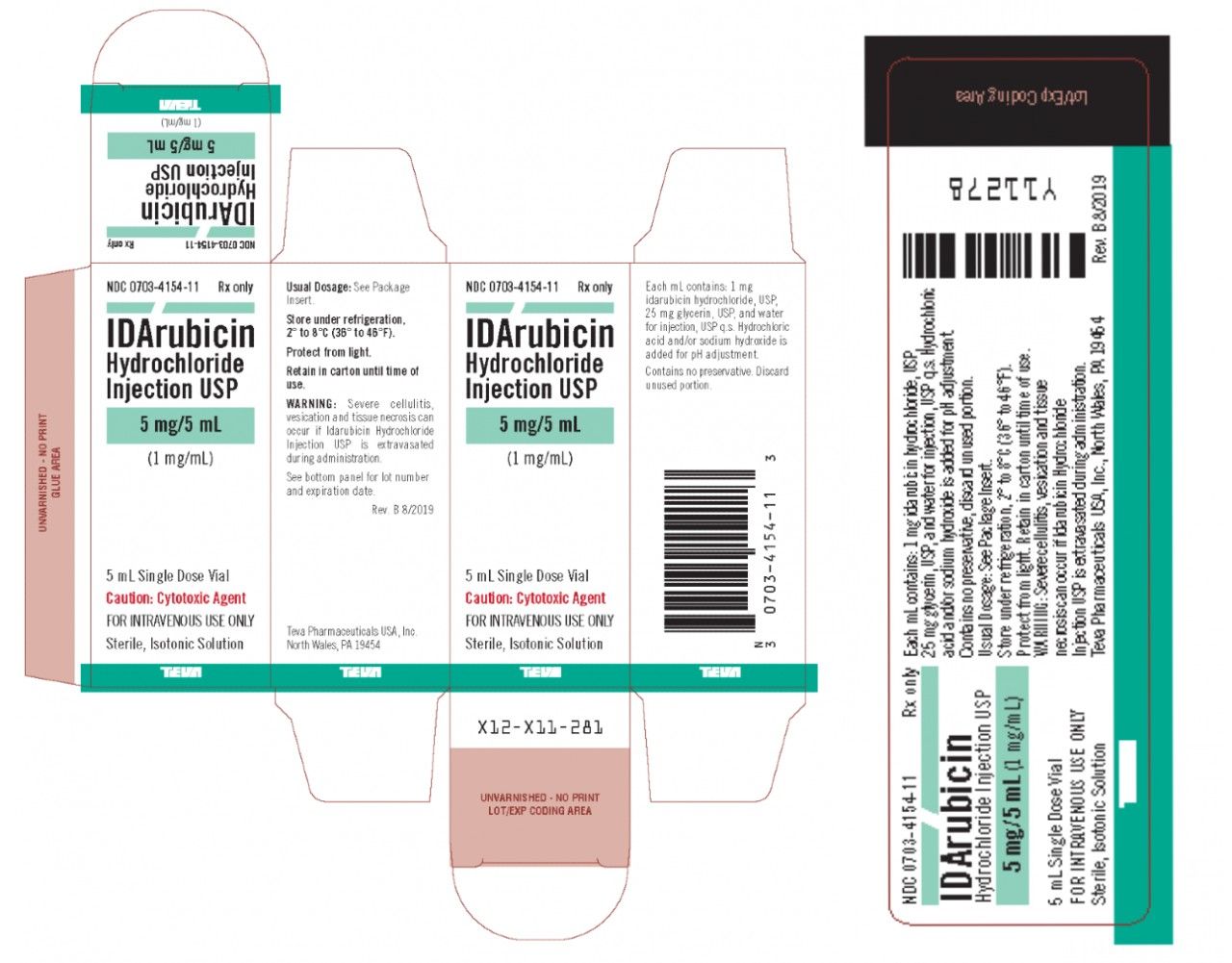
Teva Pharmaceuticals Recalls Acute Myeloid Leukemia Drug Over Particulate Matter Contamination - Top Class Actions