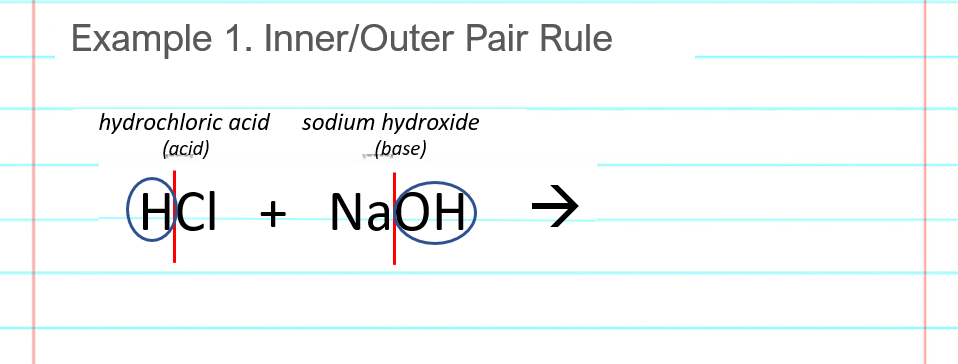
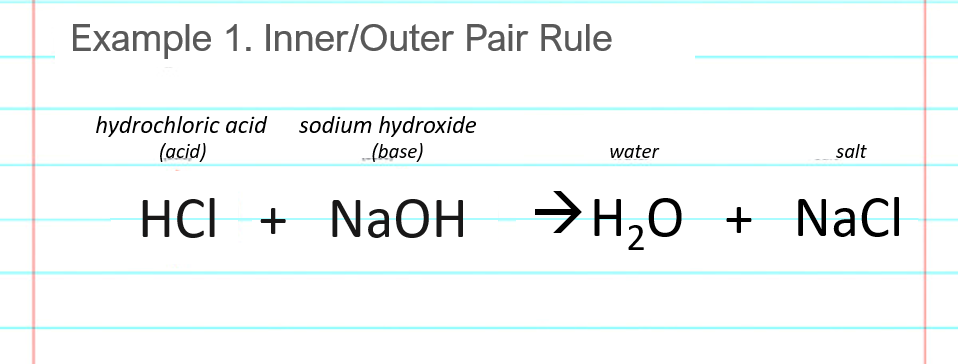
Draw the products of benzoic acid reacting with sodium hydroxide. Draw the products of the pyridine reacting with hydrochloric acid. Use the "+/-" button to add the charge (and H atom).

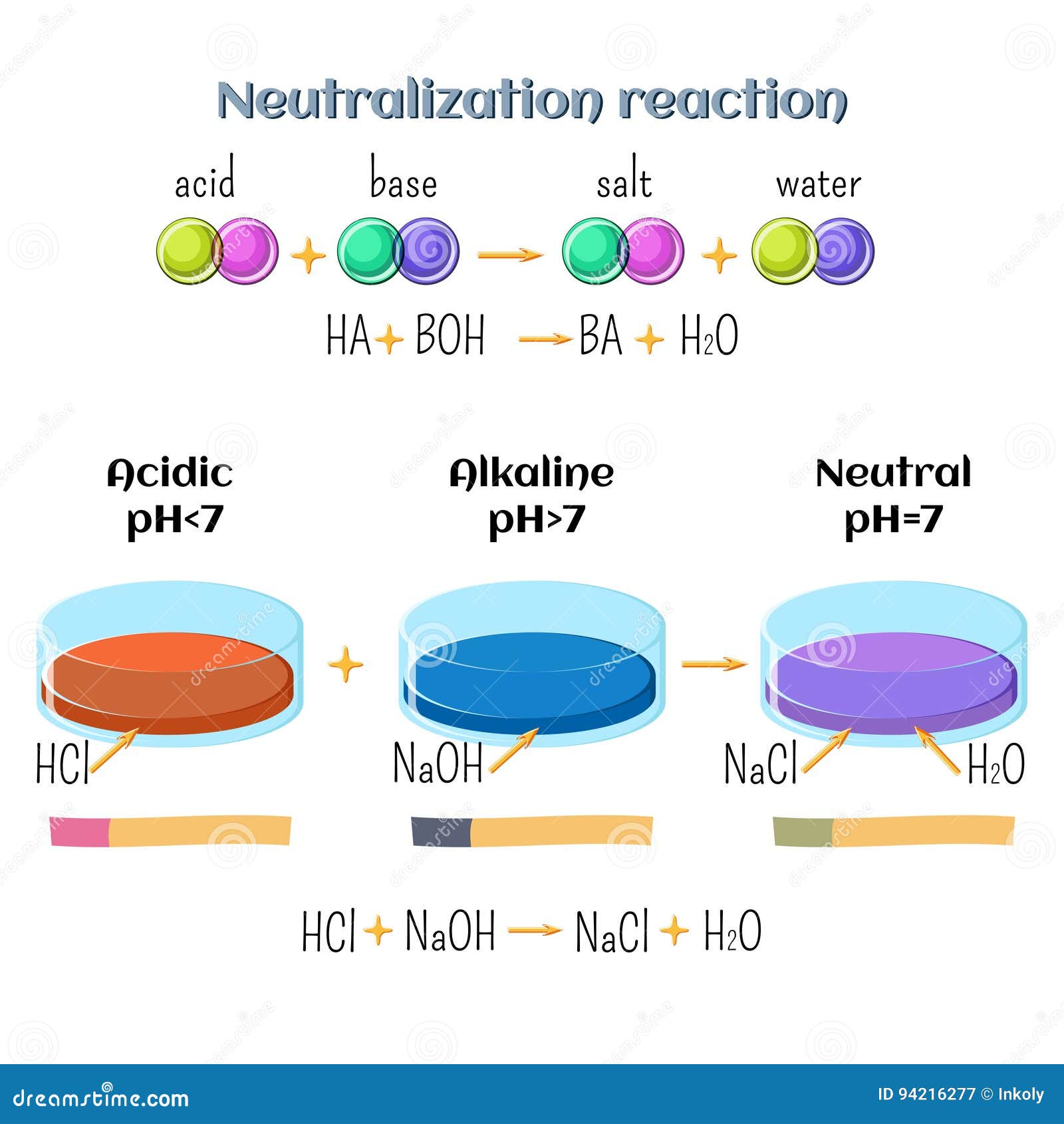
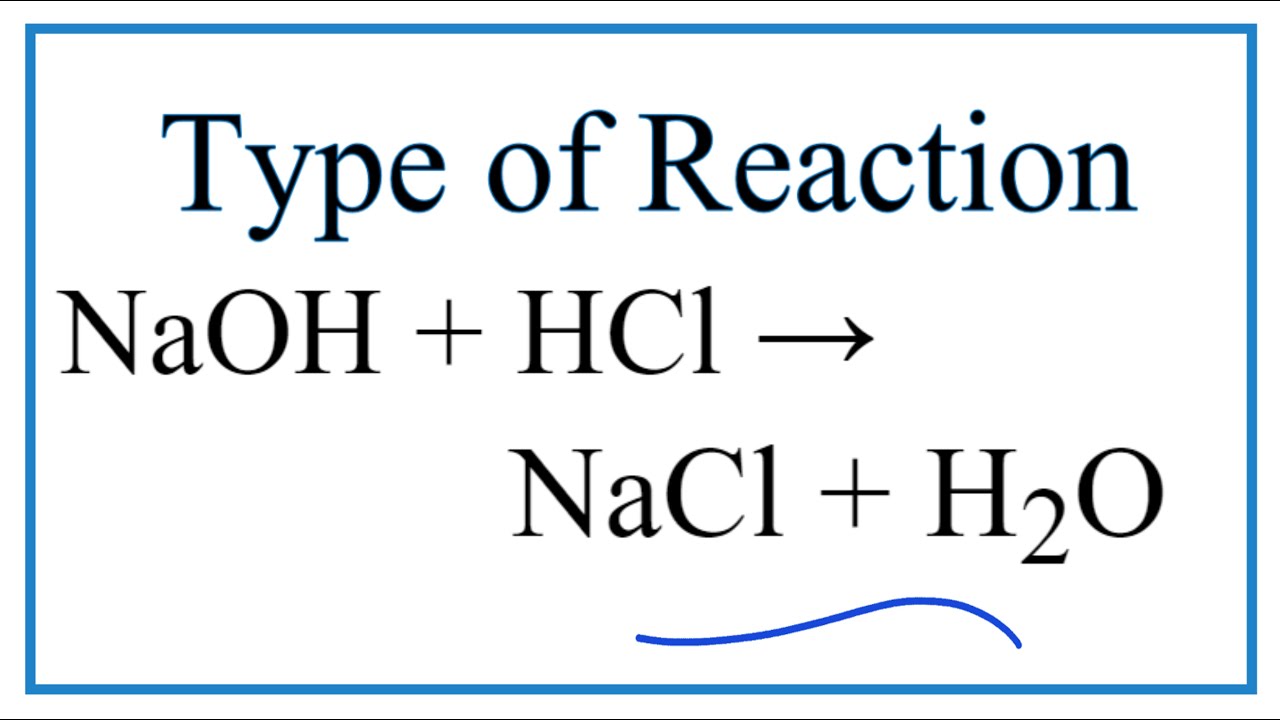
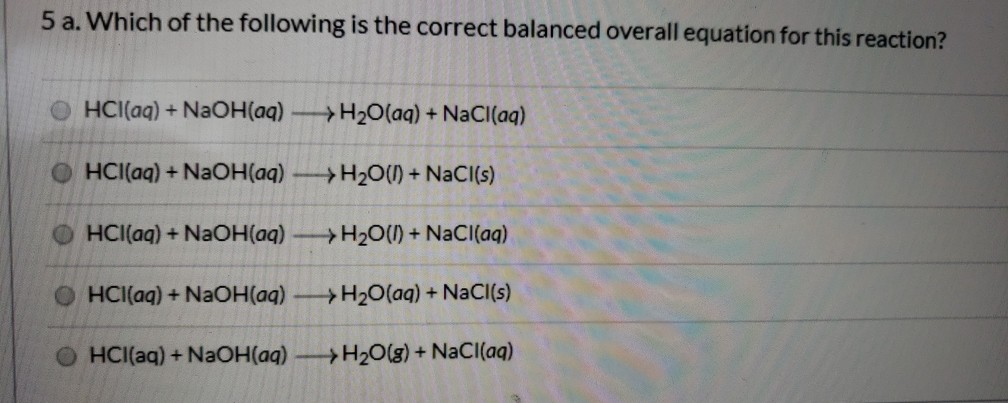
Sodium hydroxide and hydrochloric acid react as shown in this equation. NaOH ((aq))+HCl((aq))toNaCl((aq))+H(2)O((I)) (i)Which type of chemical reaction in this? (ii)The reaction is exothermic .Explain what that means. (iii)Differentiate exothermic ...

Aqueous Reactions Acids There are only seven strong acids: Hydrochloric (HCl) Hydrobromic (HBr) Hydroiodic (HI) Nitric (HNO 3 ) Sulfuric (H 2 SO 4 ) Chloric. - ppt download

SOLVED: Data and Calculation Table (20 pts) Part Acid Numbers of drops of based used in Sample Sample 2 Average 2.0 mL of 1.0 M hydrochloric phenolphthalein phenolphthalein acid drops and 18

Ionic equations A chemical equation shows the number of atoms and molecules of the reactants and products. Also shows physical state of reactants and products. - ppt download