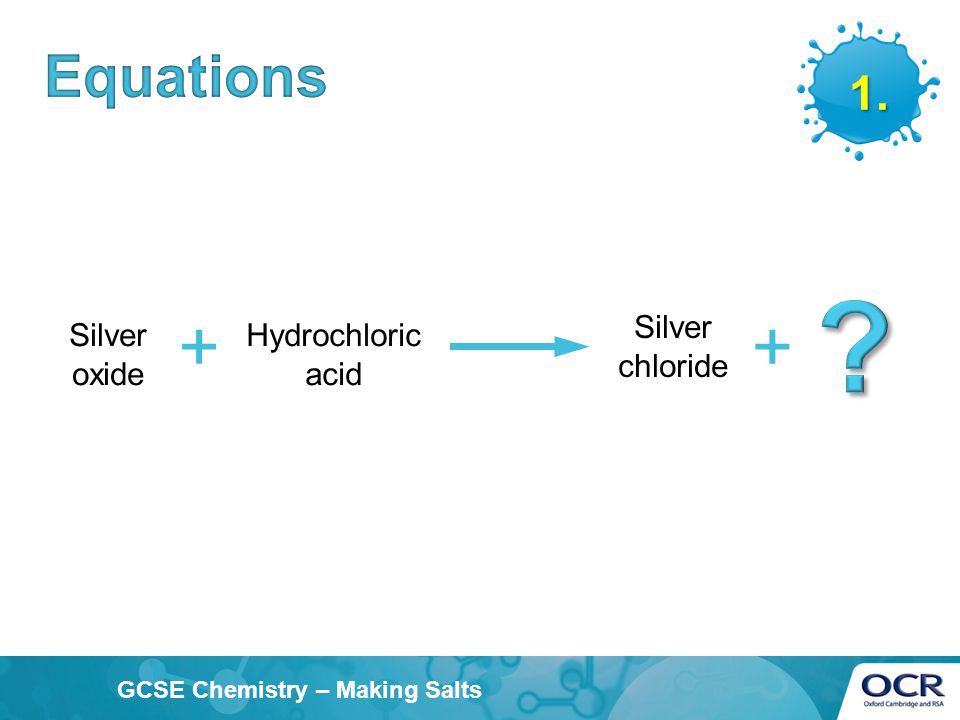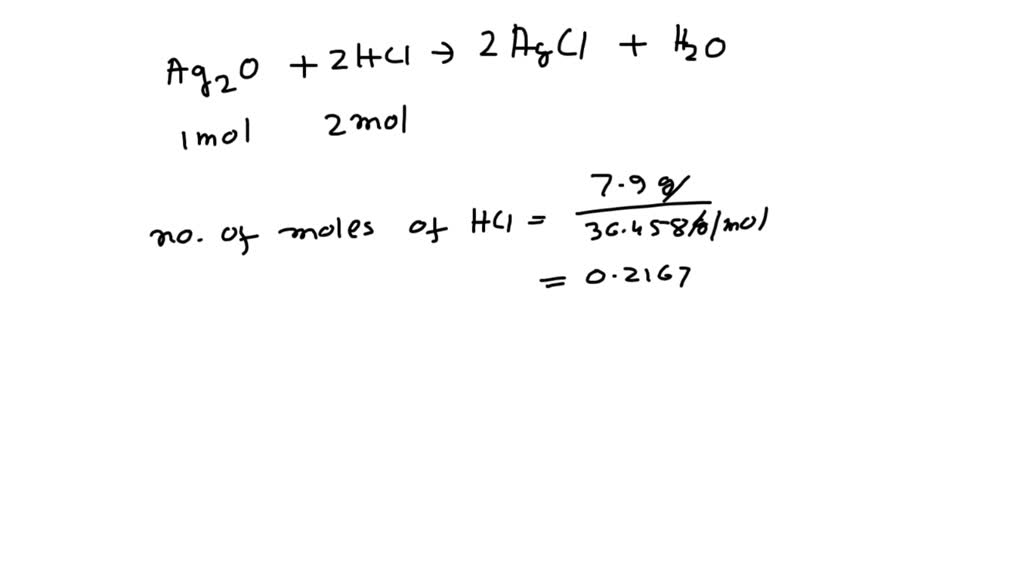
SOLVED: How many grams of silver oxide are needed to react with 7.9 g of hydrochloric acid produce silver chloide and water?

PDF) Synthesis and Characterization of Silver Oxide and Silver Chloride Nanoparticles with High Thermal Stability

PDF) Synthesis and Characterization of Silver Oxide and Silver Chloride Nanoparticles with High Thermal Stability

SOLVED: For nukstiong X9420,reod coch paxrone and write the #kelcton equation trat matchcs below: Silver Oxide reacts wlth Sodium Chloride t0 form Silver Chloride and Sodium Oxide Potasslum lodlde and Lead (IlJ

Sodium chloride (NaCl) reacts with silver nitrate (AgNO3) to produce silver chloride (AgCl) and sodium nitrate (NaNO3). The correct balanced chemical equation is :

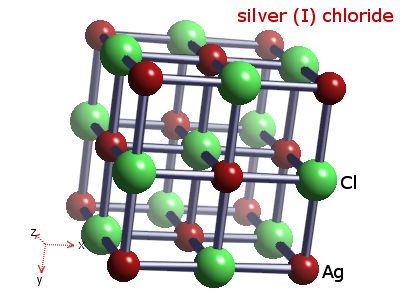
Statement: The chemical formula for silver chloride is AgCl .State whether the given statement is true or false.