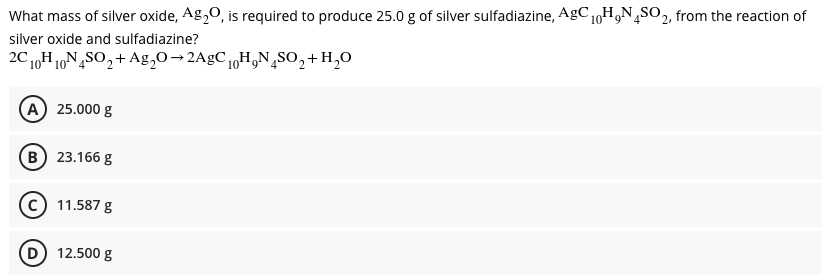
SOLVED:What mass of silver oxide, Ag2 O, is required to produce 25.0 g of silver sulfadiazine, AgC10 H9 N4 SO2, from the reaction of silver oxide and sulfadiazine? 2 C10 H10 N4

Silver oxide (Ag2O) decomposes at temperature 300^@C yielding matellic silver and oxygen gas. A 1.60g sample of impure silver oxide yields 0.104g of oxygen gas. What is the per cent by mass

Silver oxide `(Ag_2O)` decomposes at temperature `300^C` yielding matellic silver and oxygen gas... - YouTube
SOLVED: Challenge yowrself mnetal, forms an oxide with the formula M,Os This oxide contains 65.2 % of the metal by mass What'$ the Jdentity of the metal? Hint: To figure out the
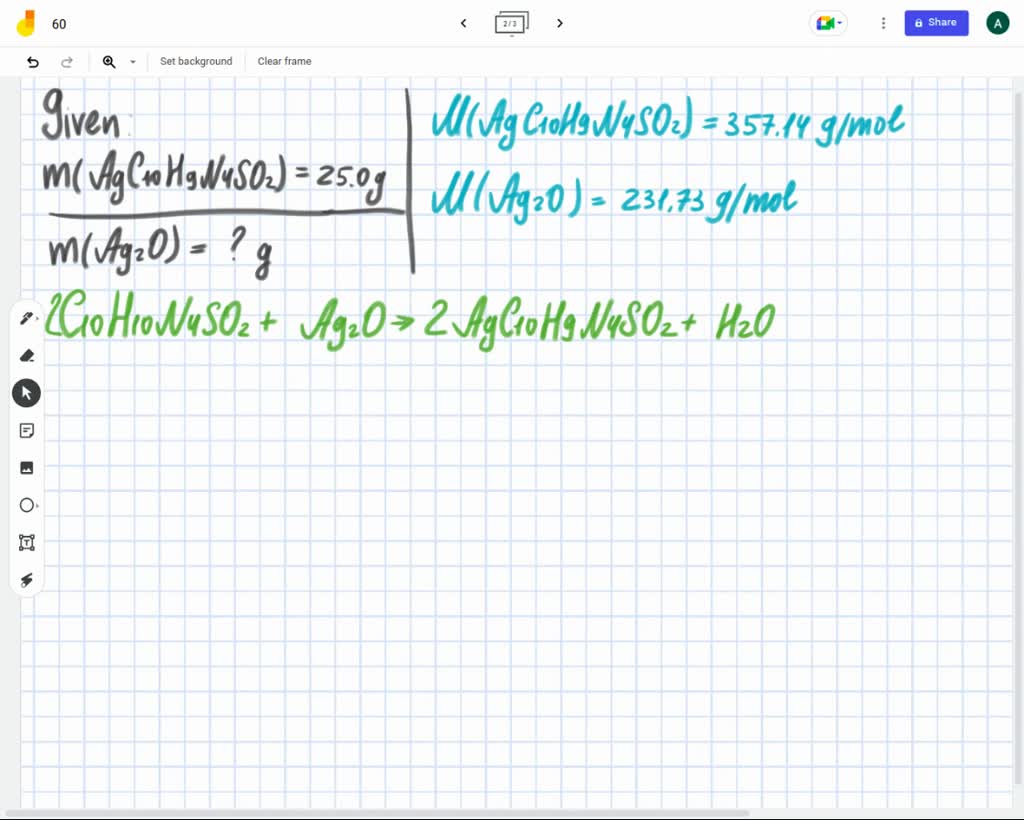
SOLVED: What mass of silver oxide, Ag2O, is required to produce 25.0 g of silver sulfadiazine, AgC10H9N4SO2, from the reaction of silver oxide and sulfadiazine?2C10 H10 N4 SO2 + Ag2 O ⟶