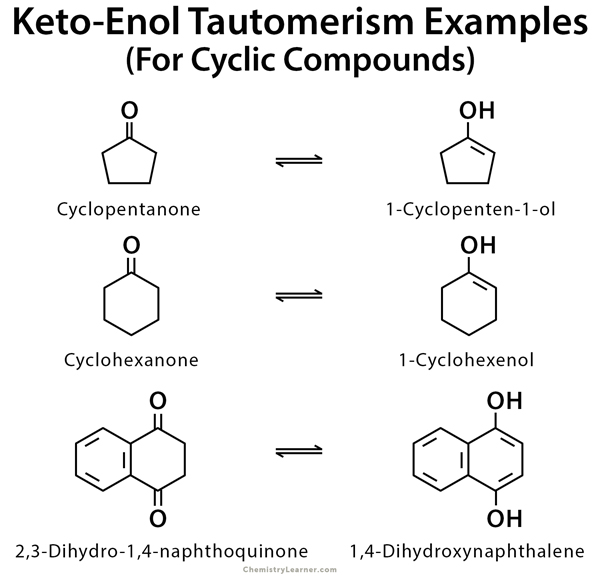
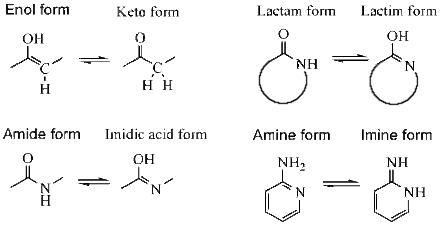
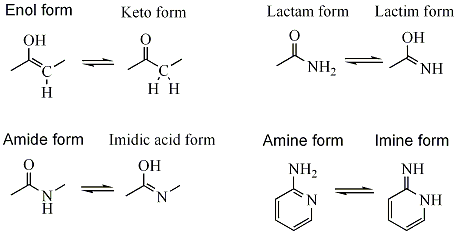
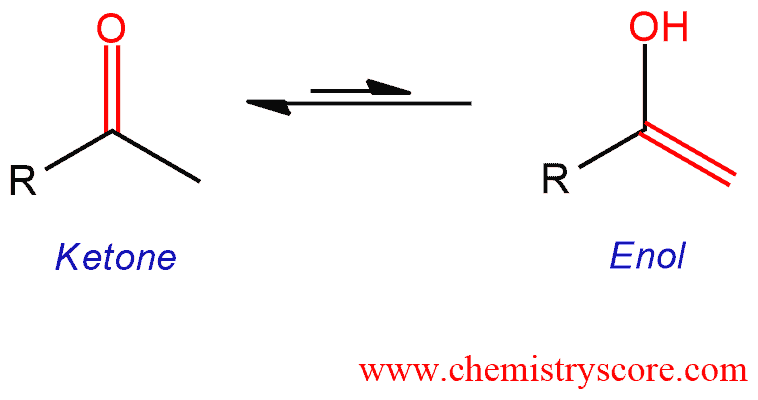
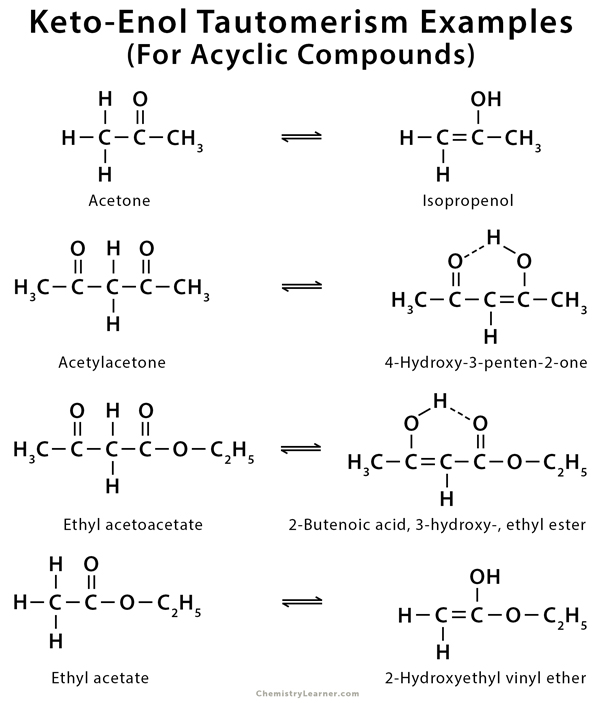
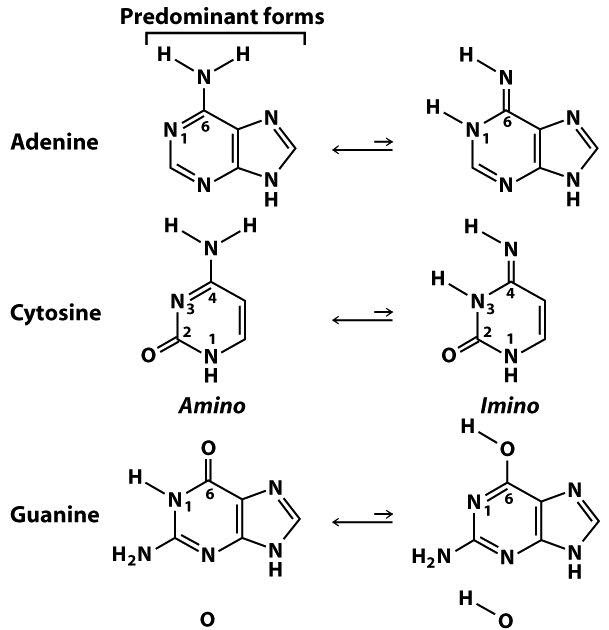
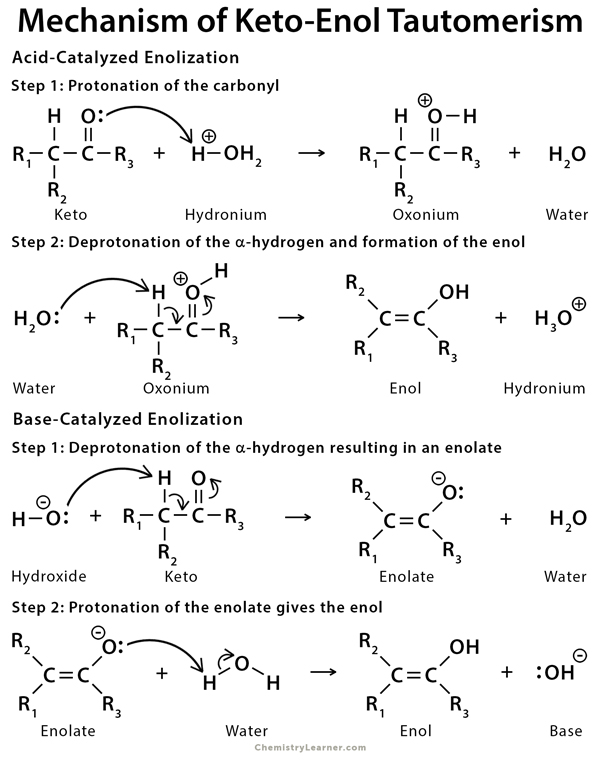
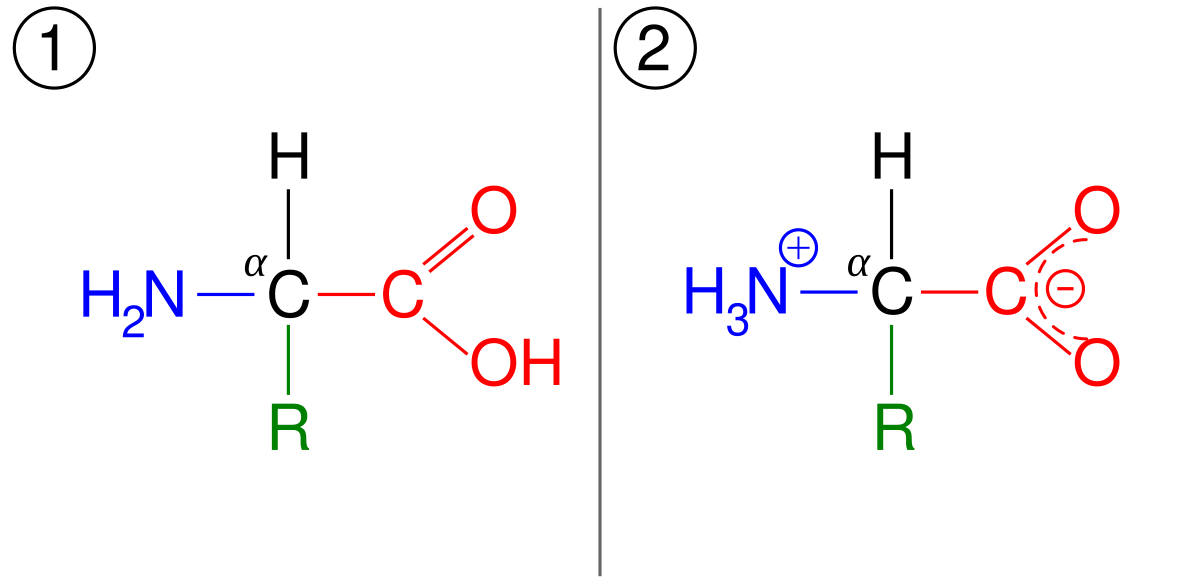
Imine–Amine Tautomerism vs Keto–Enol Tautomerism: Acceptor Basicity Dominates Over Acceptor Electronegativity in the ESIPT Process through a Six-Membered Intramolecular H-Bonded Network | The Journal of Physical Chemistry A

Molecules | Free Full-Text | Synthesis, Spectroscopic Studies and Keto-Enol Tautomerism of Novel 1,3,4-Thiadiazole Derivative Containing 3-Mercaptobutan-2-one and Quinazolin-4-one Moieties

Keto–enol tautomeric equilibrium of isoguanosine. The proportion of the... | Download Scientific Diagram