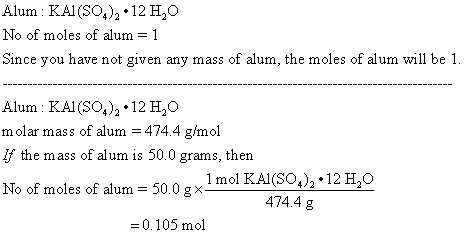Kal(so4)2.12h2o Aluminium Potassium Sulfate Potassium Alum Powder - Buy Potassium Alum Powder,Aluminium Potassium Sulfate Powder,Kal(so4)2 12h2o Potassium Alum Product on Alibaba.com

Anomalous longitudinal acoustic phonon and elastic constant in potassium alum KAl(SO4)2·12H2O single crystal studied by Brillouin spectroscopy - ScienceDirect

Kal(so4)2 12h2o Aluminium Potassium Sulfate Potash Alum Price - Buy Potash Alum Price,Aluminium Potassium Sulfate Price,Kal(so4)2 12h2o Product on Alibaba.com

Kal(so4)2 12h2o Potash Alum Potassium Alum Stone - Buy Potassium Alum Stone,Potash Alum Stone,Kal(so4)2 12h2o Potassium Alum Stone Product on Alibaba.com

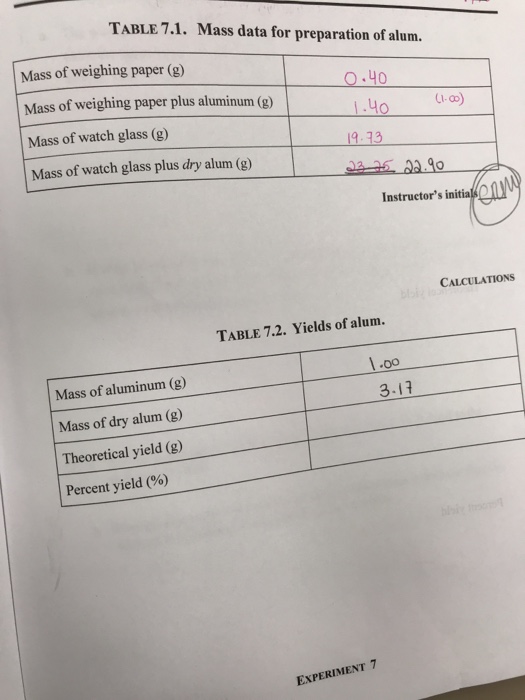
SOLVED: To synthesize alum, KAl(SO4)2 • 12H2O, 1.39 g of solid aluminum are reacted with excess potassium hydroxide and sulfuric acid as described in the experimental section of the writeup in the

Kal(so4)2.12h2o Aluminium Potassium Sulfate Potassium Alum Powder - Buy Potassium Alum Powder,Aluminium Potassium Sulfate Powder,Kal(so4)2 12h2o Potassium Alum Product on Alibaba.com
Cubic structure of alum K(Al,Cr)(SO 4 ) 2 ⋅12H 2 O, space group Pa3 _ .... | Download Scientific Diagram
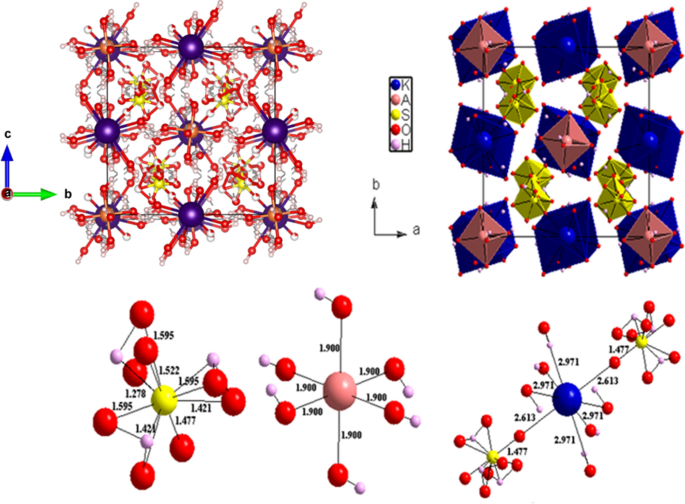
Investigations on KAl(SO4)2∙12H2O: A Candidate α-Alum Material for Energy Storage Applications | SpringerLink
Experimental Investigation and Kinetic Modeling of Potassium Alum Dodecahydrate Thermal Decomposition
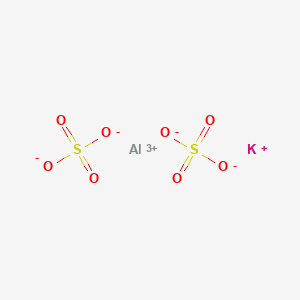
Potash alum is K Al(SO4)2. 12H2O. As a strong eletrolyte, it is considered to be 100% dissociated into K^+, Al^(3+) and SO4^(2-). The solution is acidic because of the hydrolysis of Al^(3+),