Experimental Investigation and Kinetic Modeling of Potassium Alum Dodecahydrate Thermal Decomposition

Kal(so4)2.12h2o Aluminium Potassium Sulfate Potassium Alum Powder - Buy Potassium Alum Powder,Aluminium Potassium Sulfate Powder,Kal(so4)2 12h2o Potassium Alum Product on Alibaba.com

Kal(so4)2.12h2o Aluminium Potassium Sulfate Potassium Alum Powder - Buy Potassium Alum Powder,Aluminium Potassium Sulfate Powder,Kal(so4)2 12h2o Potassium Alum Product on Alibaba.com

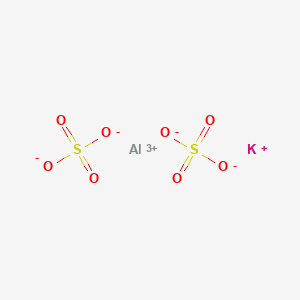
Why in KAl(SO4)2 , the oxidation no Is +3 - Chemistry - Redox Reactions - 13288081 | Meritnation.com

Potash alum is K Al(SO4)2. 12H2O. As a strong eletrolyte, it is considered to be 100% dissociated into K^+, Al^(3+) and SO4^(2-). The solution is acidic because of the hydrolysis of Al^(3+),

Cubic structure of alum K(Al,Cr)(SO 4 ) 2 ⋅12H 2 O, space group Pa3 _ .... | Download Scientific Diagram






![Potassium Alum [KAl(SO4)2] 99.7% ACS Grade Powder 1.5 Lb in Three Bottles USA 637230030875 | eBay Potassium Alum [KAl(SO4)2] 99.7% ACS Grade Powder 1.5 Lb in Three Bottles USA 637230030875 | eBay](https://i.ebayimg.com/images/g/0IMAAOSwEgVWQ-yg/s-l1600.jpg)