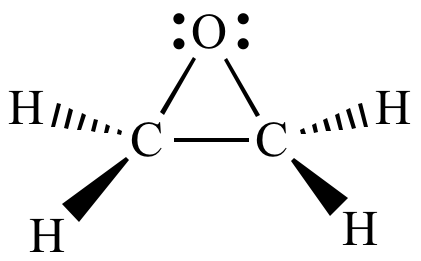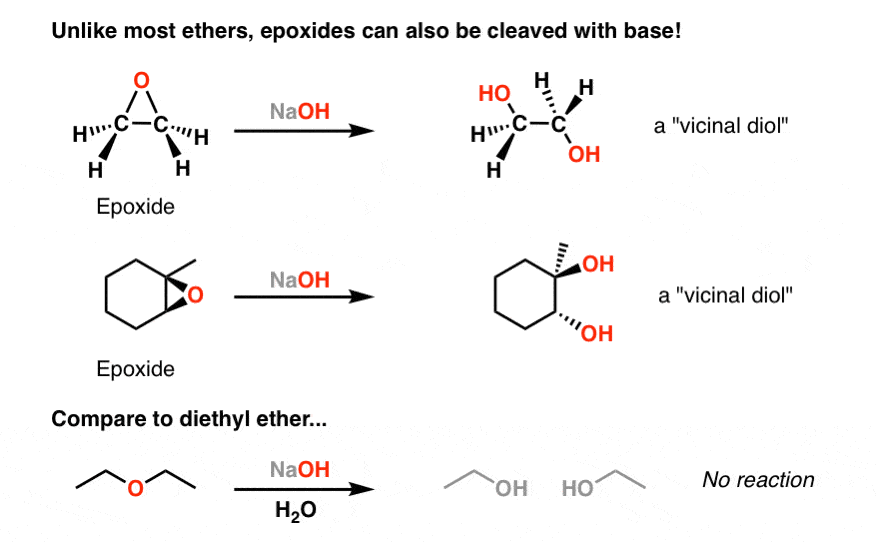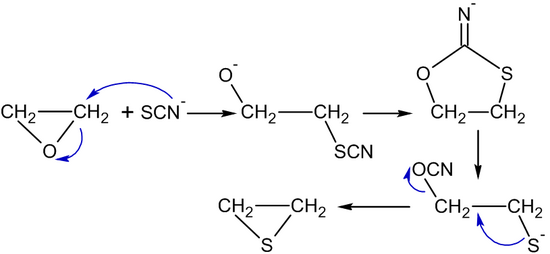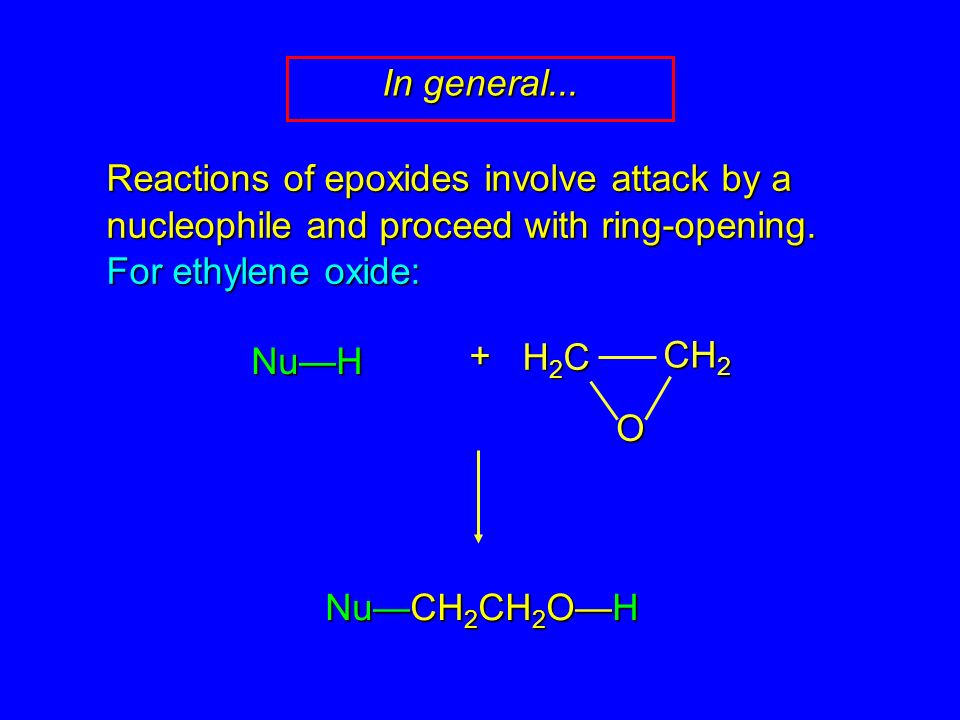
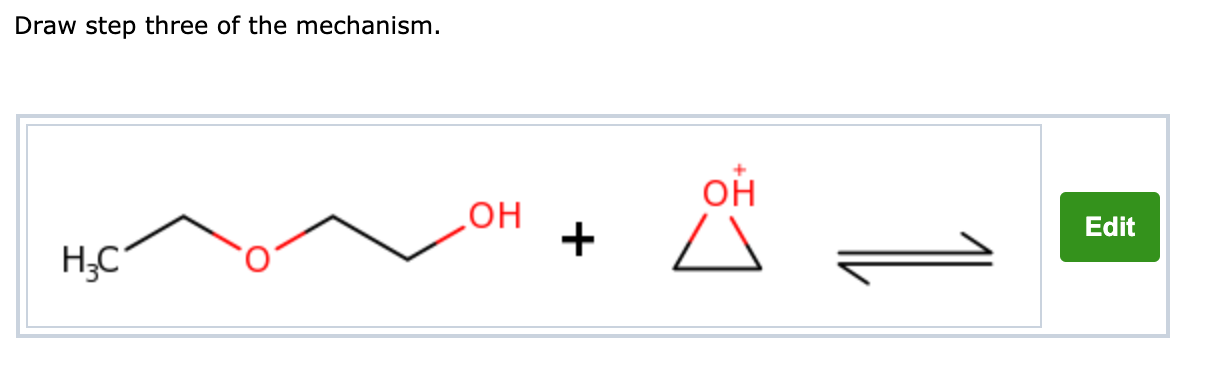
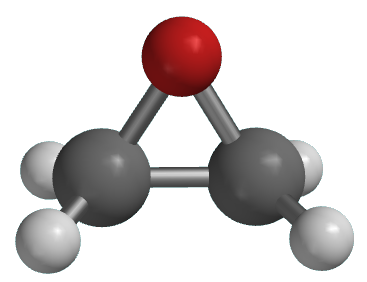
Reactions of epoxides involve attack by a nucleophile and proceed with ring-opening. For ethylene oxide: Nu—H + Nu—CH 2 CH 2 O—H H2CH2CH2CH2C CH 2 O In. - ppt download

What is oxidation of ethene in organic chemistry? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium
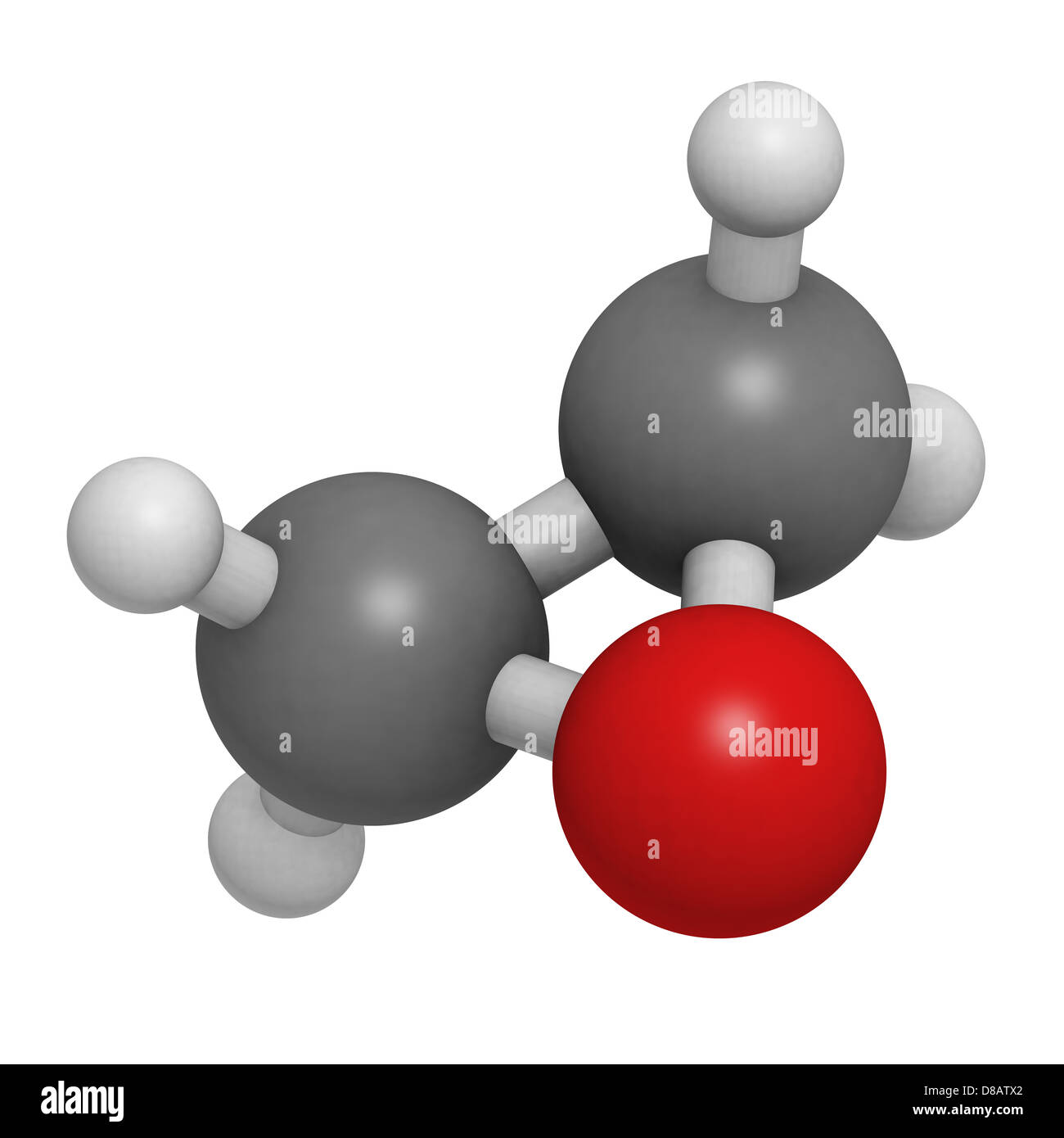
Ethylene oxide (oxirane), molecular model. Ethylene is the simplest epoxide and is used as a disinfectant and polymer precursor Stock Photo - Alamy

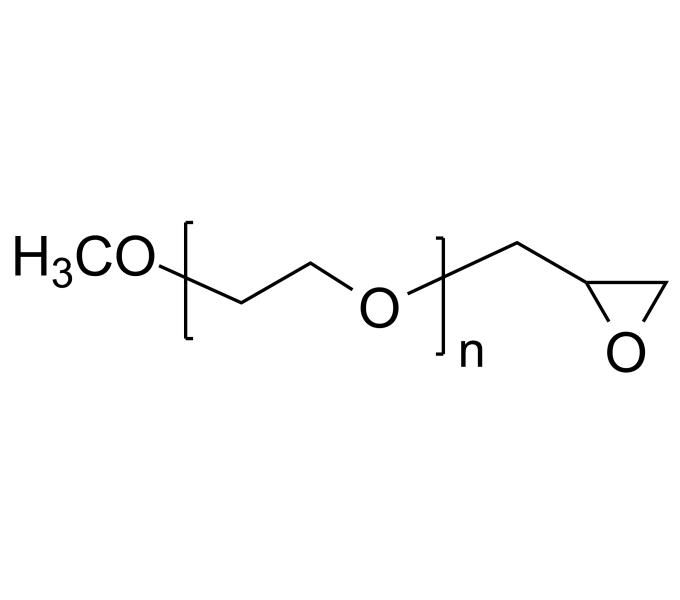

.jpg)
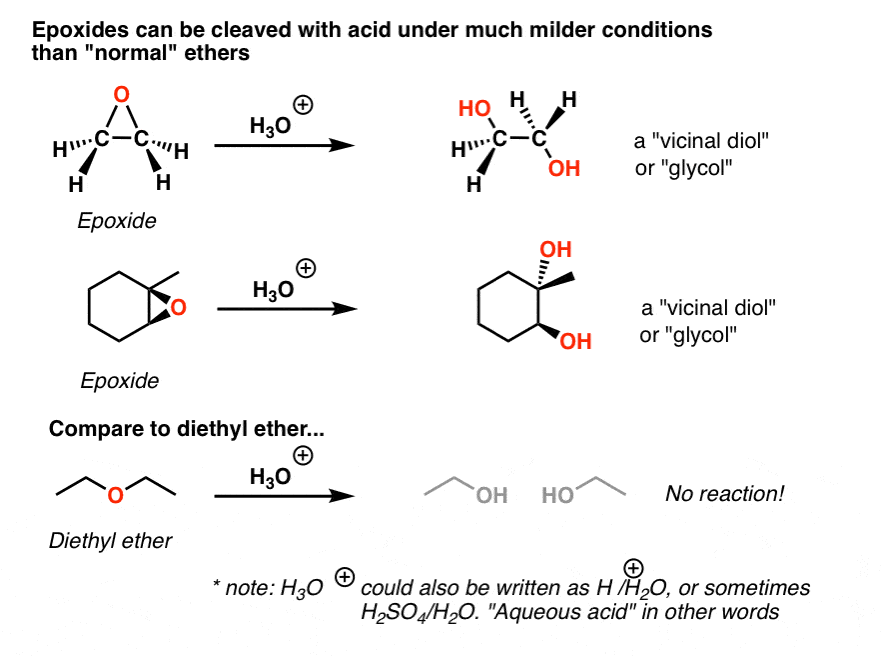


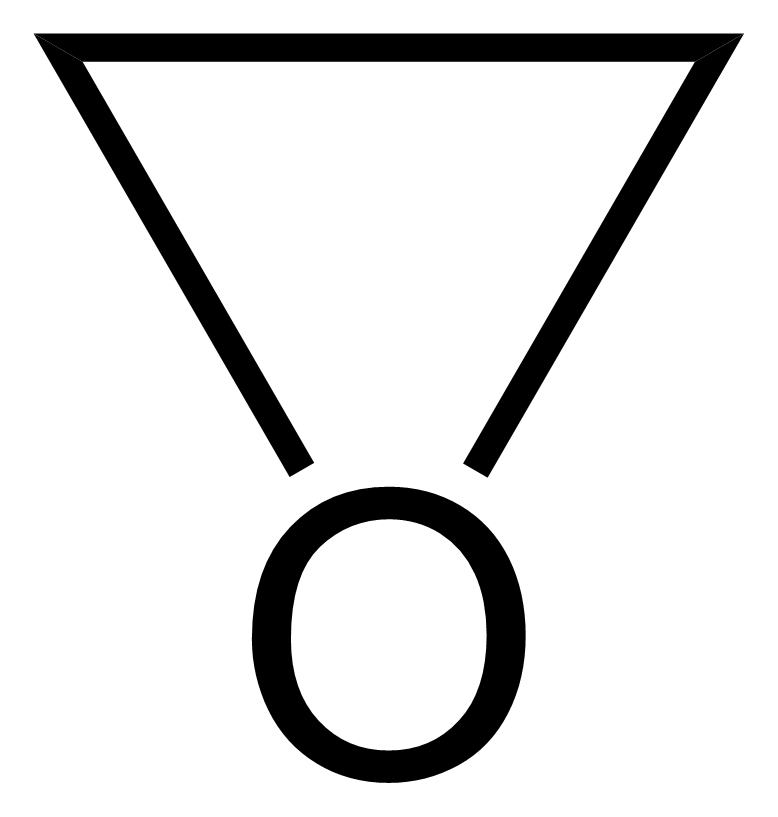

.jpg)