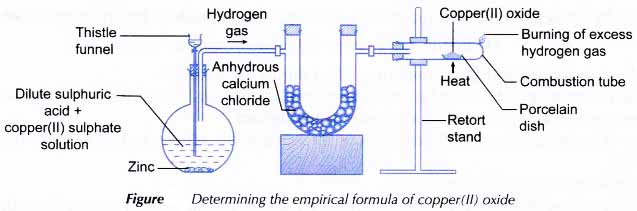
The Empirical Formula of a Copper Oxide Reading assignment: Chang, Chemistry 10th edition, pp. 55-58. Goals The reaction of hyd

✨results day✨ on Twitter: "How to determine the empirical formula of copper(II) oxide (or any compound). http://t.co/N8EtQcXNAZ" / Twitter

✨results day✨ on Twitter: "How to determine the empirical formula of copper(II) oxide (or any compound). http://t.co/N8EtQcXNAZ" / Twitter
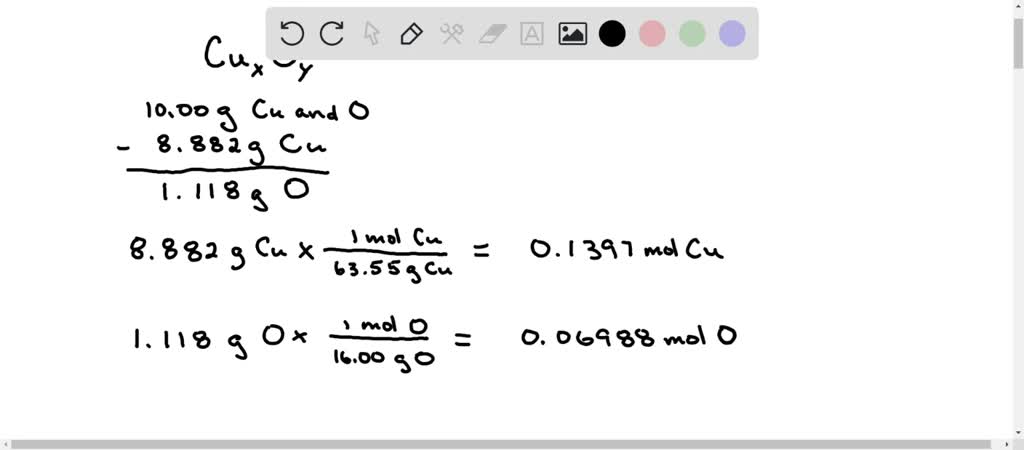
SOLVED: A 10.00 g sample of an oxide of copper was found to contain 8.882 g of copper. Determine the empirical formula of the copper oxide. Name the compound
1:36 practical: know how to determine the formula of a metal oxide by combustion (e.g. magnesium oxide) or by reduction (e.g. copper(II) oxide) - TutorMyself Chemistry

Practical: Determine the Formula of a Metal Oxide (1.5.7) | Edexcel IGCSE Chemistry Revision Notes 2019 | Save My Exams

Experimental Chemistry Emperical Formula Copper Oxide. - Lab #3 Empirical Formula Copper Oxide The - Studocu
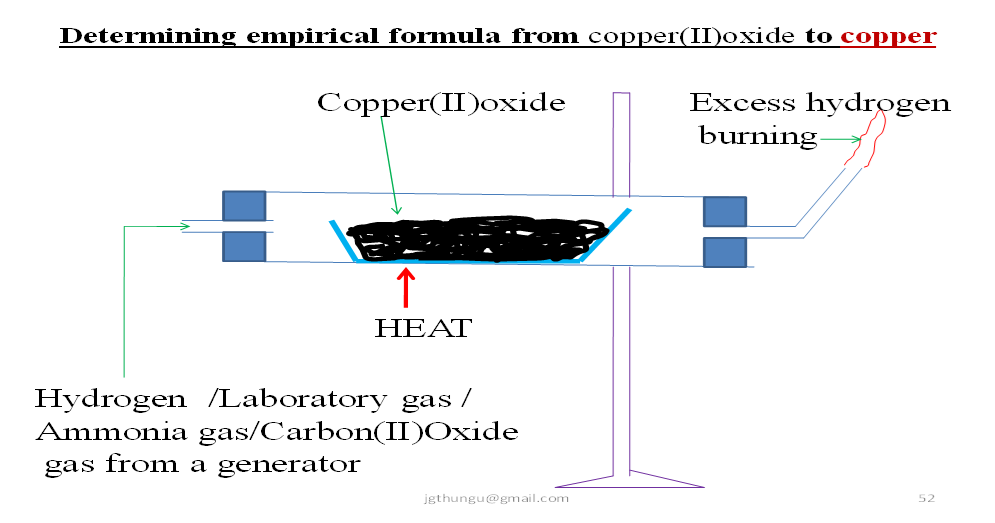
CHEMISTRY: THE MOLE-Empirical and Molecular Formula - Highschool Kenya Revision Material: 2022 Notes, Free Exams with Marking Schemes