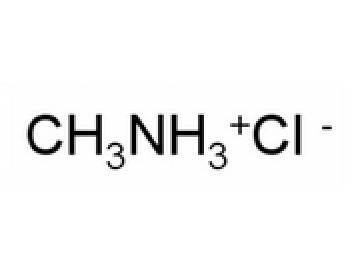
OneClass: 1. Fe(NO3)3 2. Ba(NO3)2 3. NaOCl 4. NH4I 5. NH4NO2 6. CH3NH3Cl ***I can find the correct an...

CNX Chemistry SSM Ch14 Mod06 - OpenStax College Chemistry 14: Buffers Chemistry 14: Acid-Base - Studocu

Solved) - Is the solution of KCI acidic, basic or neutral?. Is the solution... (1 Answer) | Transtutors

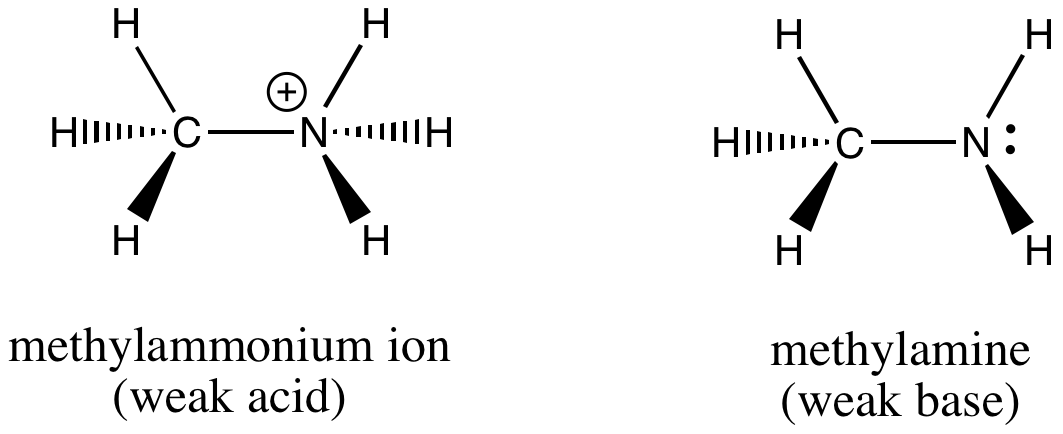
SOLVED: A buffer solution contains 0.50 M methylamine (CH3NH2) and 0.50 M methylammonium chloride (CH3NH3Cl), resulting in the equilibrium: CH3NH3+(aq) + H2O(l) ⇌ CH3NH2(aq) + H3O+(aq) One mL of 0.10 M HNO3
How would you determine is the following salts will from a solution that is acidic, basic, or pH neutral? CH3NH3CN, Fe(ClO4)3, K2CO3, CH3NH3CL, RbI | Socratic

OneClass: please write an EQUILIBRIUM reaction that shows CH3NH3Cl asacidic- Write a chemical equatio...