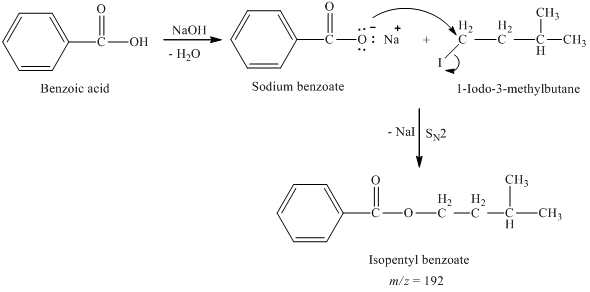
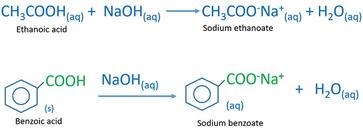
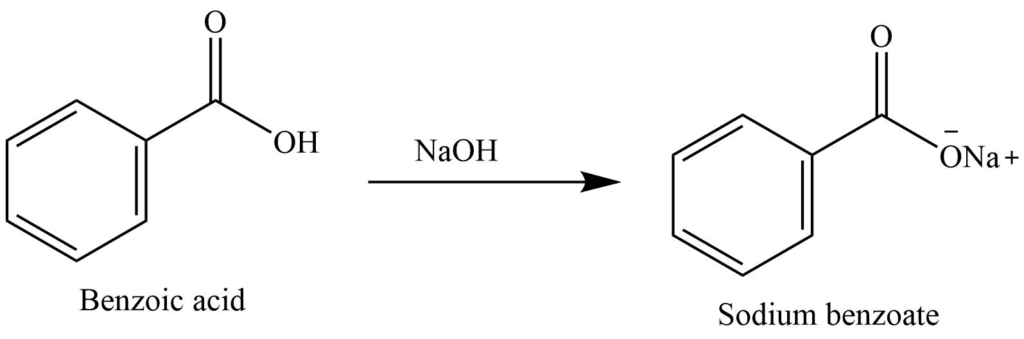
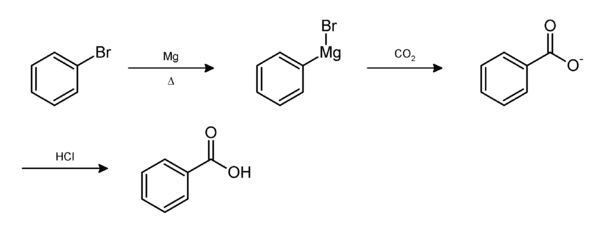
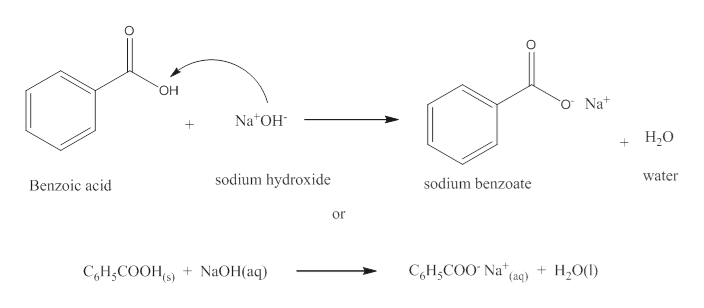
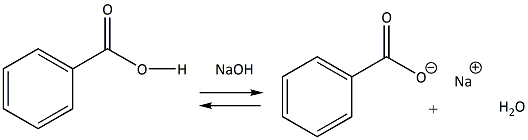
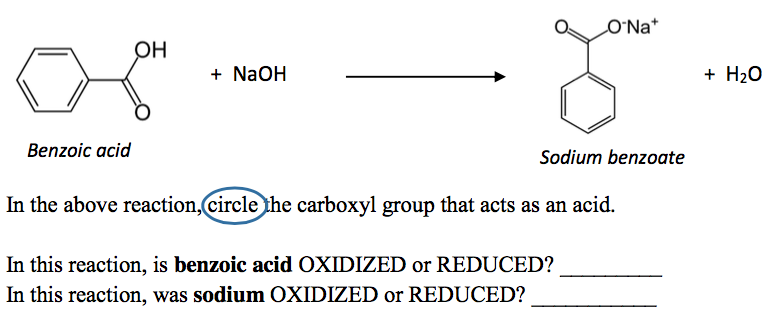Write the mechanism for the reaction of either benzoic acid or acetic acid with NaOH. Be sure to include all major structures and resonance forms. | Homework.Study.com

Draw the products of benzoic acid reacting with sodium hydroxide. Draw the products of the pyridine reacting with hydrochloric acid. Use the "+/-" button to add the charge (and H atom).

Synthesis of the internal standard 4-(methoxy-d 3 )-benzoic acid (2).... | Download Scientific Diagram

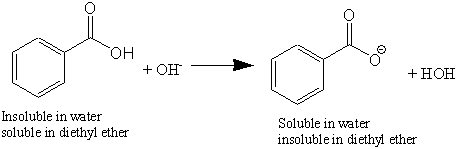
OneClass: Benzoic acid is soluble in diethyl ether but not water, however, benzoic acid is extracted ...
You are planning to purify an impure sample of benzoic acid, known to be contaminated with naphthalene. - Sarthaks eConnect | Largest Online Education Community
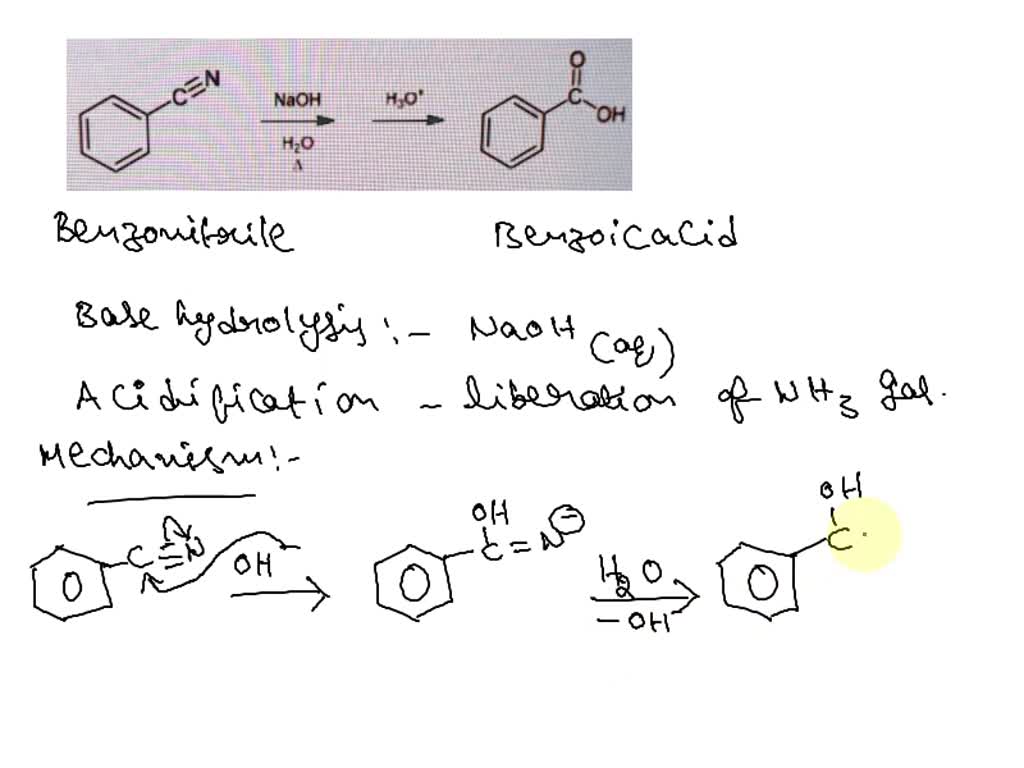
SOLVED: When benzonitrile is treated with hot; concentrated sodium hydroxide, the major product after neutralization is benzoic acid. Propose a reasonable mechanism for this reaction: (10 points) NaOH Hjo OH Hzo C=N
✓ Solved: Explain the results for the tube in which 1.0 M NaOH was added to benzoic acid. Write an equation...

Write a balanced equation for the reaction of benzoic acid with hydroxide ion. Why is it necessary to extract the ether layer with sodium hydroxide? | Homework.Study.com
![Which of the following will give phenol with \\[{\\text{CaO}}\\] and \\[{\\text{NaOH}}\\]?A. Salicylic acidB. Picric acidC. Benzoic acidD. Amino acid Which of the following will give phenol with \\[{\\text{CaO}}\\] and \\[{\\text{NaOH}}\\]?A. Salicylic acidB. Picric acidC. Benzoic acidD. Amino acid](https://www.vedantu.com/question-sets/39d15196-f3b7-4d8c-bde9-5684a8a019909186147776397460082.png)
Which of the following will give phenol with \\[{\\text{CaO}}\\] and \\[{\\text{NaOH}}\\]?A. Salicylic acidB. Picric acidC. Benzoic acidD. Amino acid