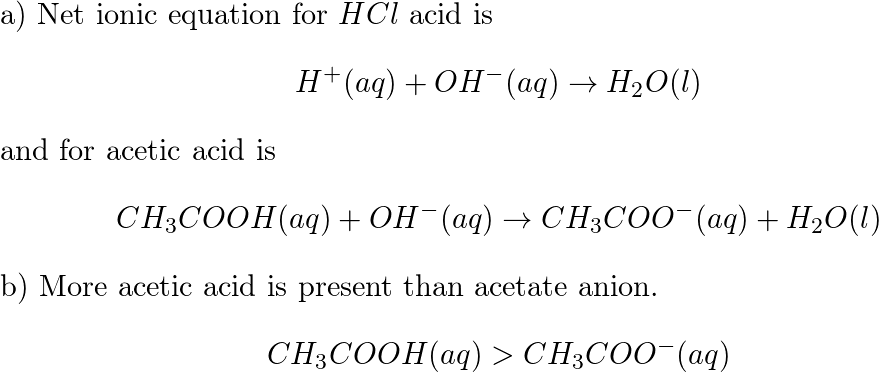How would ethanoic acid react with ammonia (NH3) and sodium hydroxide (NaOH) respectively? | Homework.Study.com
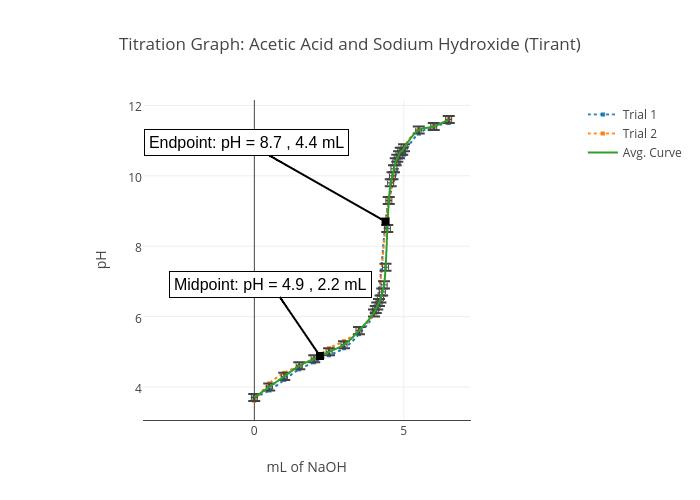
Titration Graph: Acetic Acid and Sodium Hydroxide (Tirant) | scatter chart made by Speakerrob | plotly

Lab VIII – Titration of Weak (CH3COOH) and Strong (HCL) Acids via Strong Base (NaOH) | nmiller17chem

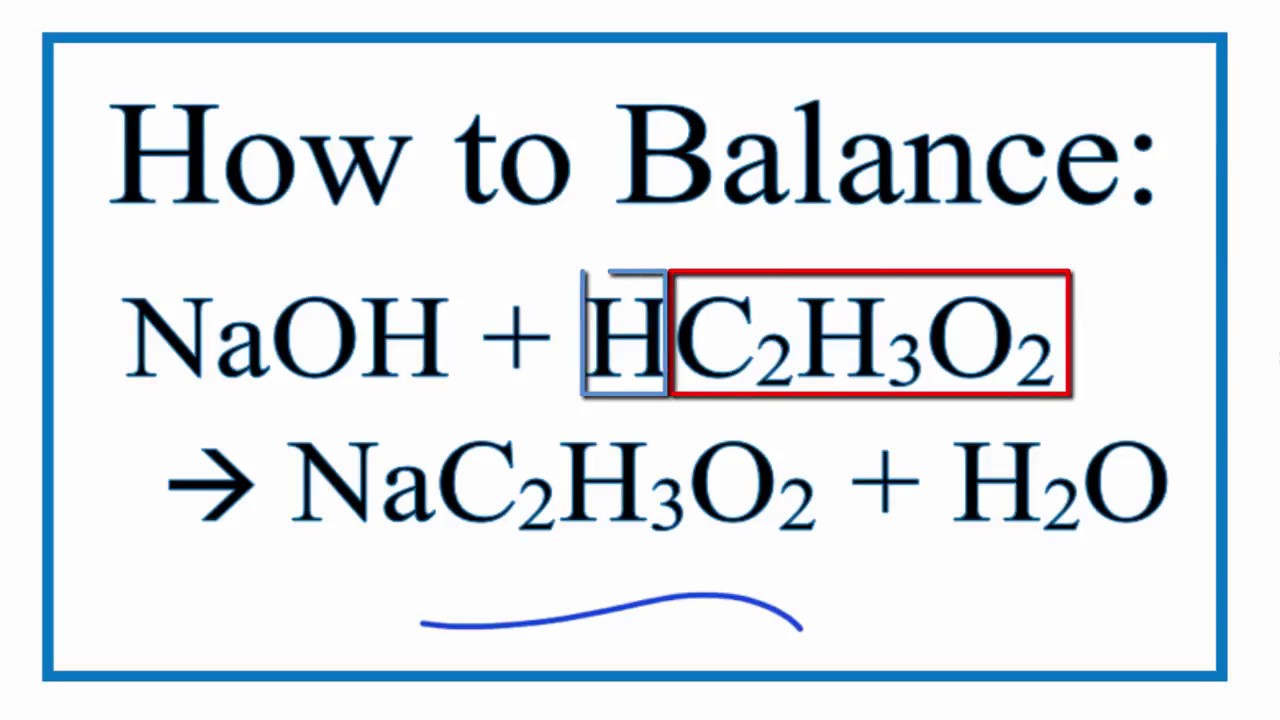
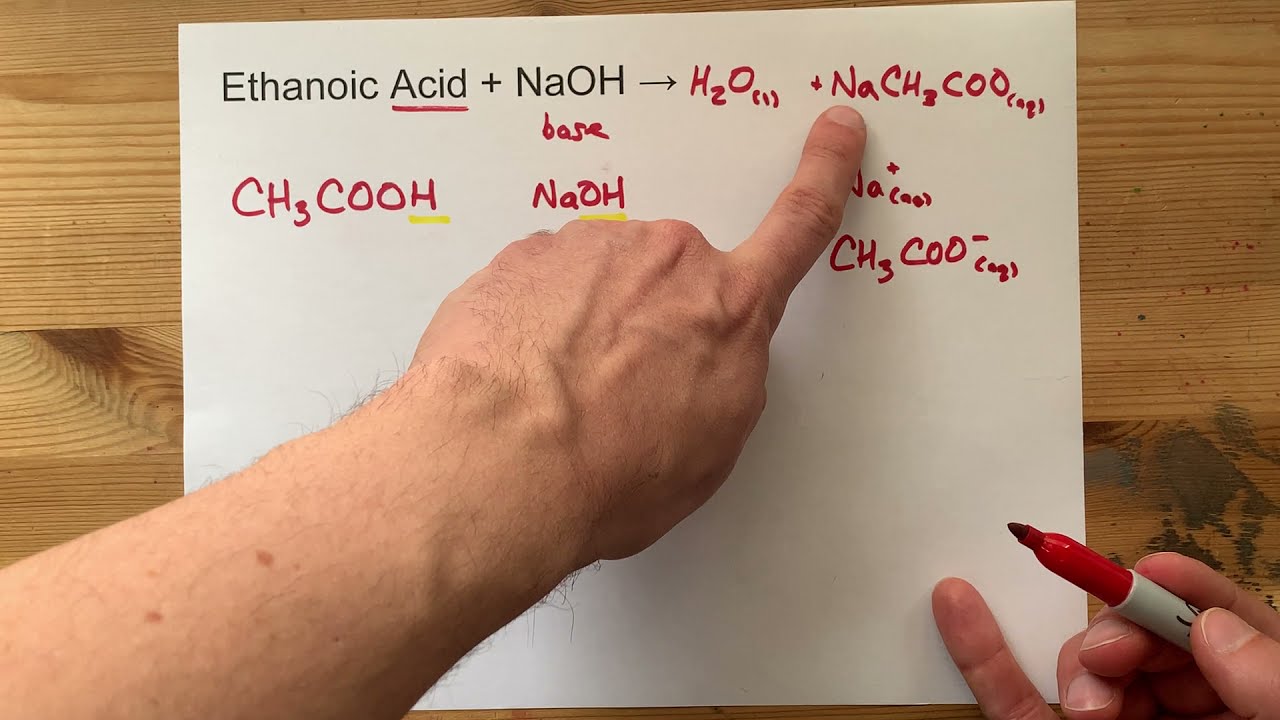
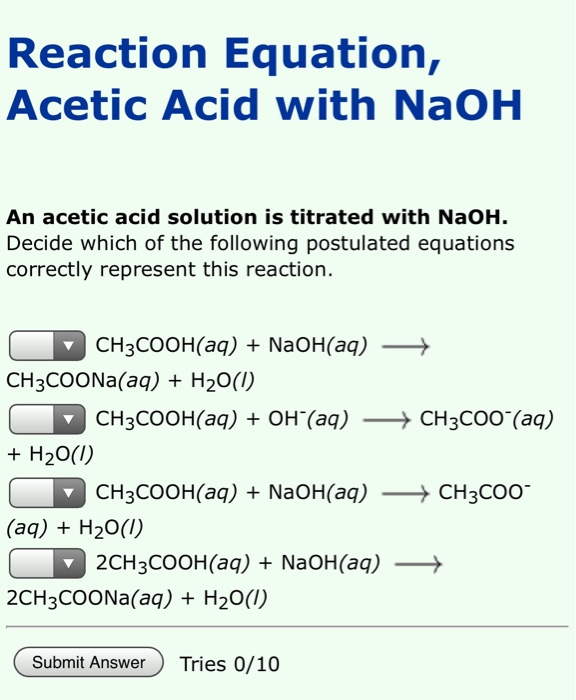
SOLVED: The balanced equation for the reaction between acetic acid and sodium hydroxide is shown below: HC2H3O2(aq) + NaOH(aq) à H2O(l) + Na C2H3O2(aq) Suppose that you perform a titration experiment to

The enthalpy of neutralization of acetic acid and sodium hydroxide is - 55.4 kJ. What is the enthalpy of ionisation of acetic acid?
Why does the solution of sodium acetate give more concentration of Hydroxide ion? Shouldn't the number of Hydroxide ion and hydrogen ion be equal? - Quora

Titration of 50 mL Acetic Acid 0.1 M with sodium hydroxide 0.1 M. (a)... | Download Scientific Diagram

SOLVED:(a) The net ionic equation for the aqueous neutralization reaction between acetic acid and sodium hydroxide is different from that for the reaction between hydrochloric acid and sodium hydroxide. Explain by writing

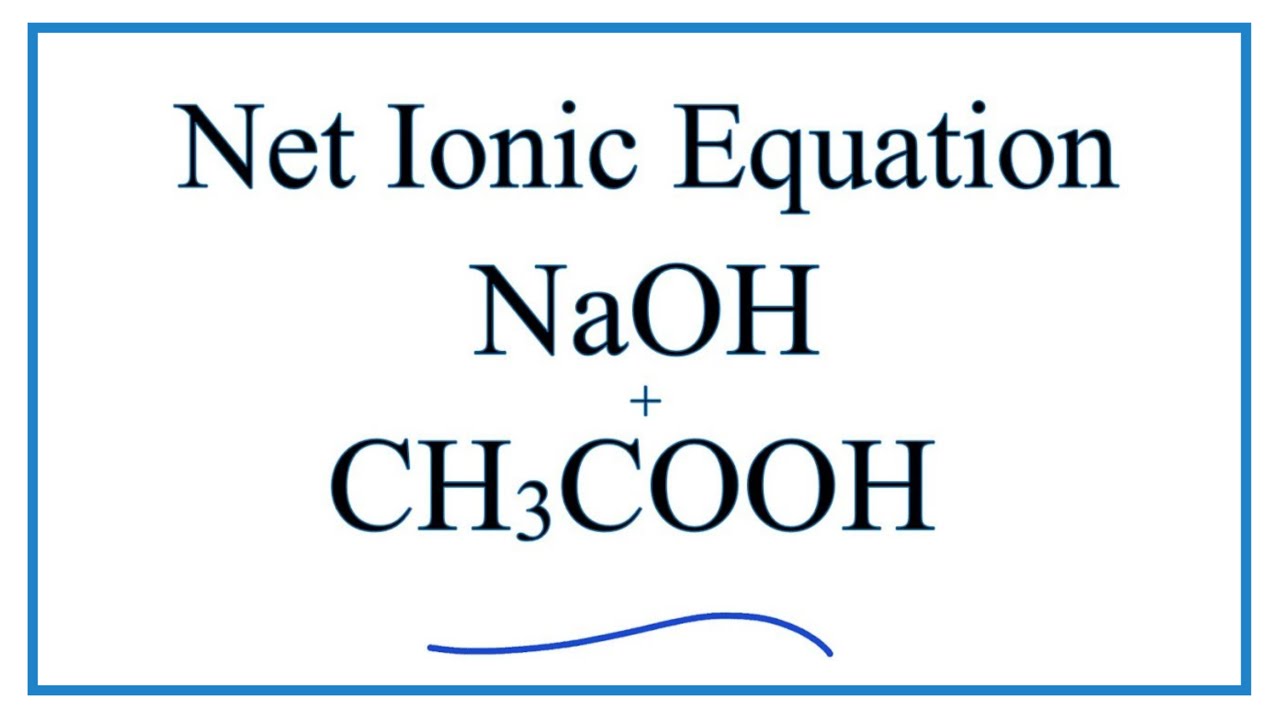
![Carbon Class 10] Complete chemical equations: (i) CH3COOC2H5 + NaOH → Carbon Class 10] Complete chemical equations: (i) CH3COOC2H5 + NaOH →](https://d1avenlh0i1xmr.cloudfront.net/82a27998-a9c5-456f-a86b-2245ca409fdb/reaction-of-ethyl-acetate-with-sodium-hydroxide---teachoo.png)