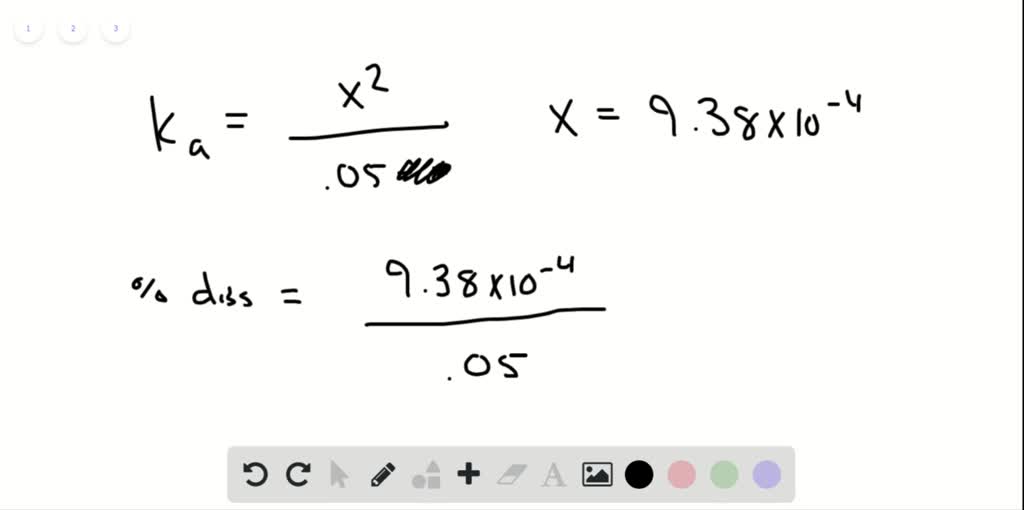
SOLVED:Calculate the percent dissociation of the acid in each of the following solutions. a. 0.50M acetic acid b. 0.050M acetic acid c. 0.0050M acetic acid d. Use Le Châtelier's principle to explain

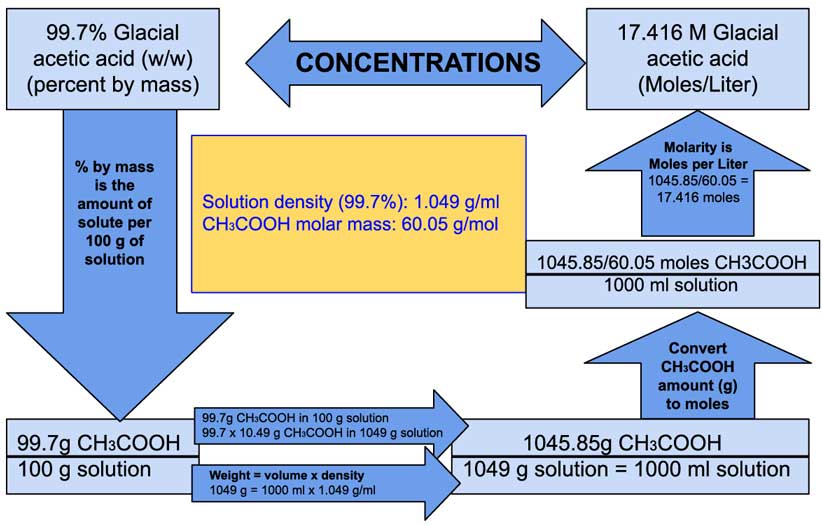
Titration What is the concentration in moles/liter of a vinegar (solution of acetic acid in water)? - ppt download
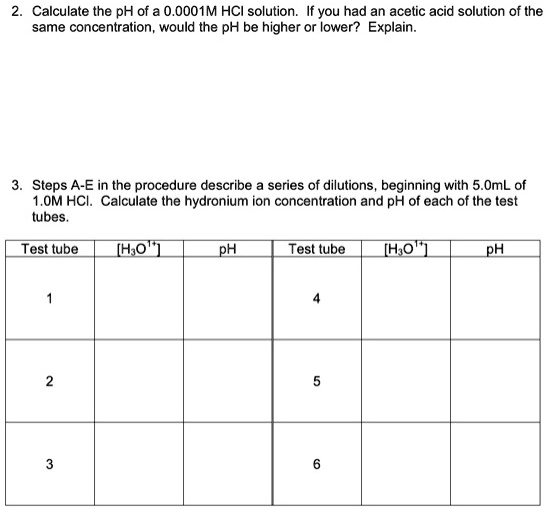
SOLVED: Calculate the pH of 0.0OO1M HCI solution If you had an acetic acid solution of the same concentration, would the pH be higher Or lower? Explain. Steps A-E in the procedure
![The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ] The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ]](https://d1hhj0t1vdqi7c.cloudfront.net/v1/QXVmVDZfQ29GV1k=/sd/)
The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ]

acid, hydrochloric acid, and citric acid. Dilution factor is calculated... | Download Scientific Diagram

What volume of a 2.5 M stock solution of acetic acid is required to prepare 100.0 milliliters of a .50 M acetic acid solution? | Socratic

Calculate the pH of a 0.01 N solution of acetic acid. K(a) for CH(3)CO OH is 1.8xx10^(-5) at 25^(@)C.
Calculate amount of acetic acid which should be added in 300ml of 0.1 Molar of acetic acid solution (ka=10^ 5) to double its (i) alpha (II) pH
The pH of an acetic acid solution is 3.26. What is the concentration of acetic acid and what is the percent of acid that's ionized? - Quora